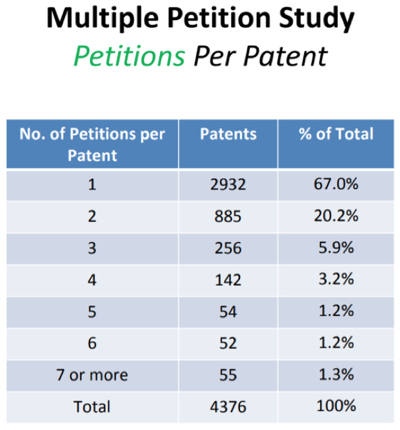
Virtual Reality Patents Survive Patent-Eligibility Challenge
By James Korenchan —
 Last week, Judge Seeborg of the U.S. District Court for the Northern District of California ruled that claims related to virtual reality applications are patent eligible under 35 U.S.C. § 101. Plaintiff Electronic Scripting Products, Inc. (ESPI) accused HTC American, Inc. (HTC) of direct and induced infringement through HTC's VIVE virtual reality headsets. Defendant HTC then brought the § 101 challenge against ESPI in a motion to dismiss. While this challenge failed, HTC was still successful in having ESPI's complaint thrown out for failure to state a claim, and the Court granted ESPI leave to amend.
Last week, Judge Seeborg of the U.S. District Court for the Northern District of California ruled that claims related to virtual reality applications are patent eligible under 35 U.S.C. § 101. Plaintiff Electronic Scripting Products, Inc. (ESPI) accused HTC American, Inc. (HTC) of direct and induced infringement through HTC's VIVE virtual reality headsets. Defendant HTC then brought the § 101 challenge against ESPI in a motion to dismiss. While this challenge failed, HTC was still successful in having ESPI's complaint thrown out for failure to state a claim, and the Court granted ESPI leave to amend.
The patents at issue in this case, U.S. Patent No. 8,553,935 (the '935 patent) and U.S. Patent No. 9,235,934 (the '934 patent), are in the same family and are directed to measuring pose (i.e., a three-dimensional (3D) position) and orientation of objects in real 3D environments using on-board photodetectors and stationary light sources. The claimed methods and apparatuses have both virtual reality and augmented reality applications. Both patents state a need in the art for improved motion tracking of all six degrees of freedom inherent in 3D space, which can be achieved with more robust and rapid pose determination of objects, such as remotes, wands, pointers, and gaming controllers. The tracked pose and motion of such objects can be digitized with respect to a reference location in real space, which makes one-to-one motion mapping between real space and cyberspace possible. Ideally, such improvements can reduce or eliminate frequency-encountered problems with a user's quality of experience, such as information loss, position aliasing, gradual drift, and position error.
Representative claims of each patent are as follows:
U.S. Patent No. 8,553,935:
12. A system comprising a manipulated object, said system comprising:
a) a first plurality of predetermined light sources disposed at known positions in world coordinates;
b) a photodetector mounted on-board said manipulated object for generating light data indicative of light detected from said first plurality of light sources;
c) a relative motion sensor mounted on-board said manipulated object for generating relative motion data indicative of a change in an orientation of said manipulated object; and
d) a processor for determining the pose of said manipulated object based on said light data and said relative motion data, wherein said pose is determined with respect to said world coordinates.
U.S. Patent No. 9,235,934:
1. A wearable article cooperating with a first plurality of predetermined light sources disposed in a known pattern, said wearable article comprising:
a) a photodetector configured to detect said first plurality of predetermined light sources and generate photodetector data representative of the positions of said first plurality of predetermined light sources; and
b) a controller configured to identify a derivative pattern of said first plurality of predetermined light sources from said photodetector data, wherein said derivative pattern is indicative of the position of said photodetector.
In hopes of dismissing these claims on Alice grounds, HTC asserted that the claims are directed to patent-ineligible abstract concepts of observing visible points in space and determining the position (e.g., the pose and orientation) of an object relative to the viewer. HTC went as far as characterizing the claims as an attempt to patent an abstract process of using objects to determine one's position, "as sailors have done by looking at the stars for centuries." Further, HTC cited to 2016's In re TLI Communications LLC Patent Litigation, in which the Federal Circuit found the recited tangible components of a telephone unit and a scanner to amount to no more than conduits for an abstract idea of "classifying an image and then storing the image based on its classification." HTC argued that the tangible components here — namely, the recited photodetector and the motion sensor — are likewise such conduits.
In response, ESPI relied almost entirely on the Federal Circuit decision in Thales Visionix Inc. v. U.S., in which the Federal Circuit found patent-eligible claims that were "not merely directed to the abstract idea of using mathematical equations for determining the relative position of a moving object to a moving reference frame," but rather "directed to systems and methods that use inertial sensors in a non-conventional manner to reduce errors in measuring the relative position and orientation of a moving object on a moving reference frame." Further, ESPI noted various manners in which the asserted patents provide novel advantages over conventional systems, such as by increasing the speed and accuracy of 3D pose recovery, reducing noise, and reducing system complexity, and cost.
HTC attempted to distinguish ESPI's claims from those in Thales, arguing that, while the eligible claims in Thales provided an improvement over the prior art by having a specific configuration of elements and a particular arrangement of sensors, ESPI's claims do not require any specific configuration of either the recited photodetectors or light sources, and also do not provide an improvement over the prior art.
However, the Court agreed that the claims were similar enough to those in Thales to survive under Alice (albeit saying it was a "close call"), saying that HTC's attempts to distinguish this case from Thales mischaracterize what ESPI's patents state. Addressing step one of the Alice framework, the Court highlighted differences between the patents and conventional methods. While conventional methods use photodetectors positioned in and around the environment in order to determine a manipulated object's pose, ESPI's patents specify that a remote control or the manipulated object itself is equipped with motion sensors and the photodetector. The Court then went on to further emphasize the improvements and non-conventionality of the claimed invention, noting that ESPI recognized a need for improved motion tracking in virtual reality applications "and accomplished this by reversing the usual placement of markers and sensors and using only light sources," thus providing a novel arrangement "eliminat[ing] the need for multiple synchronized imaging cameras located in the environment and also minimiz[ing] the bandwidth and processing needs of the system."
Concluding its short step one analysis, the Court echoed ESPI's arguments with respect to Thales, and distinguished the claims from those in TLI Communications, stating that the recited sensors "are not mere conduits for abstract principles, but instead their placement is integral to the improved functioning of the system."
Rather than stop its analysis there, the Court quickly addressed step two, stating that the claims would have survived regardless of the outcome of step one. Briefly, citing BASCOM Global Internet Services, Inc. v. AT&T Mobility LLC for support ("[A]n inventive concept can be found in the non-conventional and nongeneric arrangement of known, conventional pieces."), the Court yet again emphasized the claimed invention's simpler, less expensive, versatile, and non-conventional approach, calling it "sufficiently inventive."
This Order is a positive one for patent owners in the virtual and augmented reality fields. The Court focused quite heavily here on the improvements and advantages of the claimed invention over more conventional approaches. If patent owners in these fields can invite and rely on the same focus, it would be a useful tool in withstanding Alice scrutiny.
Order Granting Motion to Dismiss by District Judge Seeborg