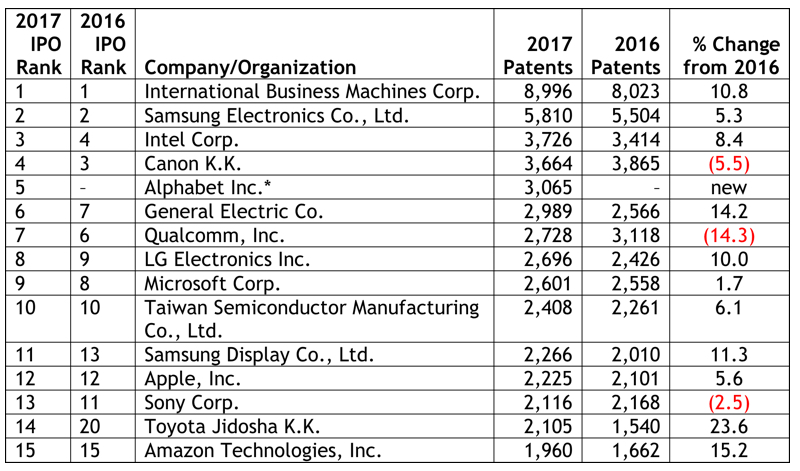
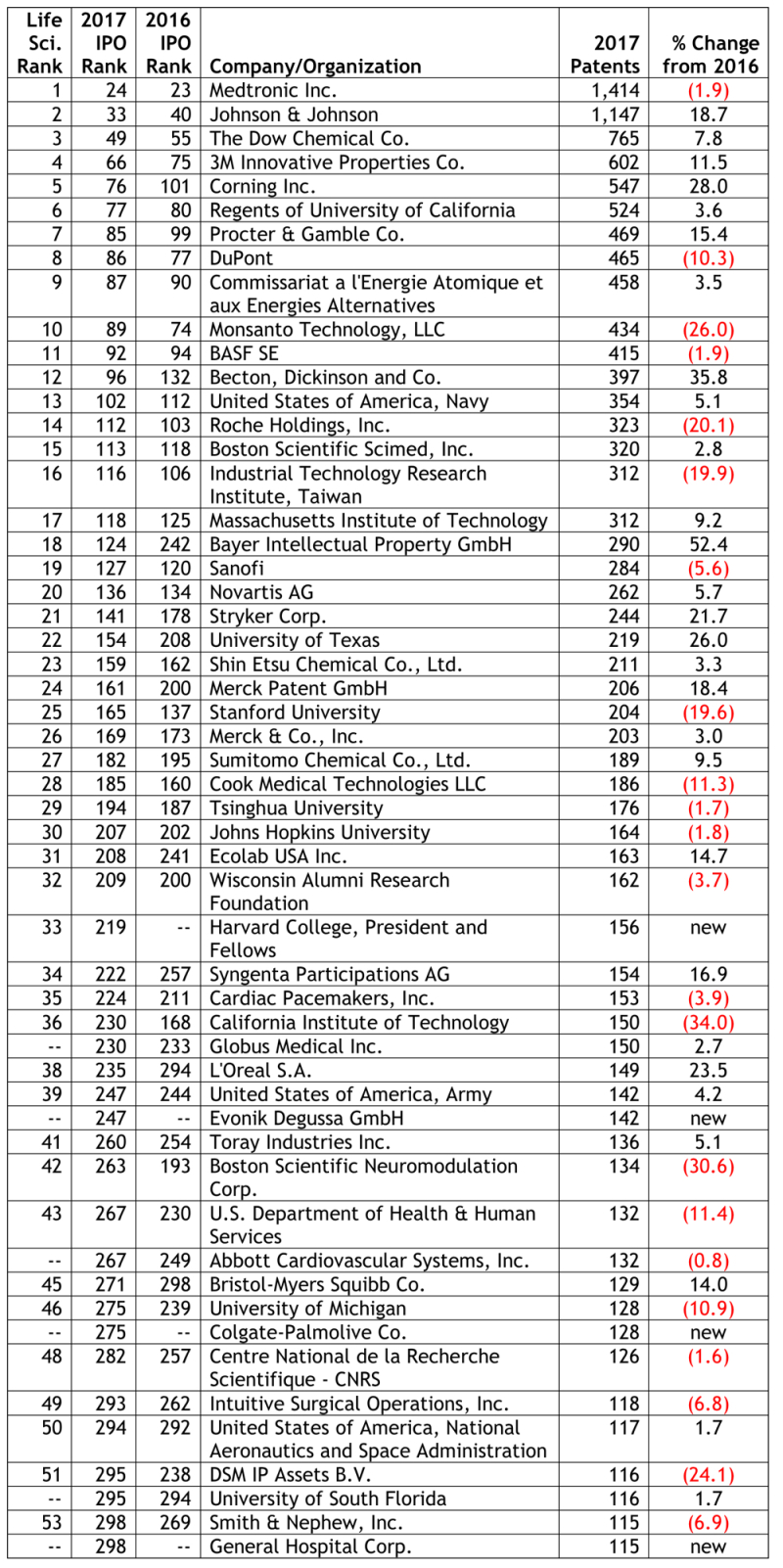
By Kevin E. Noonan –
 In the 1970's, the New York Tristate area was entertained by (or subjected to) television and radio ads for a discount stereo outlet named Crazy Eddie's. Under the tag line "Our Prices are Insane," the ads involved a number of outrageous scenarios, one of which was "Christmas in July" (or August), complete with Santa Claus in shorts hawking electronic wares.
In the 1970's, the New York Tristate area was entertained by (or subjected to) television and radio ads for a discount stereo outlet named Crazy Eddie's. Under the tag line "Our Prices are Insane," the ads involved a number of outrageous scenarios, one of which was "Christmas in July" (or August), complete with Santa Claus in shorts hawking electronic wares.
Representatives Thomas Massie (R-KY, 4th dist.) and Marcy Kaptur (D-OH, 9th dist.) introduced a bill recently that is reminiscent of Christmas in July, at least for pro-patent people. The bill, H.R. 6264, entitled "Restoring America's Leadership in Innovation Act," seeks to undo many of the changes in U.S. patent law produced by the Leahy-Smith America Invents Act of 2012. While Rep. Kaptur's interests (and bona fides) in patent law are unclear, Rep. Massie's are not. He holds a degree in electrical engineering from MIT and has been awarded two dozen patents. He has also been quoted extensively on patent law; for example, speaking of the proponents of patent "reform," on July 5, 2017:
A lot of those companies want to become automobile manufacturers, or cell phone manufacturers, or they want to write software for operating systems, but they didn't invent in those areas and they don't own the patents that have historically been the touchstone of innovation ownership. "They'd love to just come in and start playing in those fields and start using their size and scale as an advantage, and to them, patents look like a hindrance," Massie explained. "They are here in Congress looking to weaken patents and they are not just interested in weakening patents issued in the future, they are looking to weaken all patents."
"Reform is not about strengthening the patent granting system, but about making it harder for patent owners to assert patents to protect their rights," Massie said emphatically. "They didn't read the Constitution and come to say they have a better way more in line with what the Founding Fathers had in mind. They just want all the patents to be public domain today. It is very short sighted to attack the patent system in this way."
The bill has as its avowed purpose "[t]o promote the leadership of the United States in global innovation by establishing a robust patent system that restores and protects the right of inventors to own and enforce private property rights in inventions and discoveries, and for other purposes." It asserts that "[t]he Leahy-Smith America Invents Act (Public Law 112–29) enacted on September 16, 2011, and several decisions of the Supreme Court have harmed the progress of Science and the useful Arts by eroding the strength and value of the patent system" (Sec. 2(2)) and that "[a] United States patent secures a private property right to an inventor" (Sec. 2(4)).
The substantive provisions are as follows:
Sec. 3, entitled "Repeal of First-to-File System under the America Invents Act," restores U.S. patent law to a "first to invent" regime (Sec. 3(b)), and restores the meaning of "public use" and "on sale in this country" to their pre-AIA definitions (Sec. 3(c)).
Sec. 4 abolishes inter partes (IPR) and post grant review (PGR), and provides a "sense of Congress" that (1) IPR and PGR have harmed innovation by subjecting inventors to serial patent challenges; (2) that these procedures have invalidated patents at "an unreasonably high rate"; (3) that patents should not be subjected to "unfair" PTAB proceedings but should be adjudicated in court; (4) IPR, PGR, and inter partes reexamination to be repealed but (5) ex parte reexamination will remain (Sec. 4(c)).
Sec. 5 abolishes the Patent Trial and Appeal Board (PTAB) as being unnecessary in view of elimination of IPRs and PGRs. It reestablishes the Board of Patent Appeals and Interferences (BPAI) in its stead (Sec. 5(a)) and the BPAI's duties (Sec. 5(b)) and also reestablishes appeals to district court (Sec. 5(c)).
Sec. 6 eliminates fee diversion from the USPTO and establishes an "Innovation Promotion Fund" (Sec. 6(a)(5)), to be funded by any fees collected by the patent and trademark activities of the Office to be used to fund the operations of the Office. These provisions are to come into force on the first day of the first fiscal year after enactment; the bill also provides for termination of the Reserve Fund (Sec. 6(b)) "[u]pon the payment of all obligated amounts."
Sec. 7(a) relates to changes intended to overcome the effects of the Supreme Court's recent § 101 jurisprudence. This section derives from the proposals made by the IPO and AIPLA and reads as follows:
AMENDMENT.—Section 101 of title 35, United States Code, is amended to read as follows:
§ 101. Inventions patentable
(a) IN GENERAL.—Whoever invents or discovers any new and useful process, machine, manufacture, or composition of matter, or any new and useful improvement thereof, may obtain a patent therefor, subject to the conditions and requirements of this title.
(b) EXCEPTION.—A claimed invention is ineligible patent subject matter under subsection (a) if the claimed invention as a whole, as understood by a person having ordinary skill in the art, exists in nature independently of and prior to any human activity, or exists solely in the human mind.
(c) ELIGIBILITY STANDARD.—The eligibility of a claimed invention under subsections (a) and (b) shall be determined without regard as to the requirements or conditions of sections 102, 103, and 112 of this title, or the claimed invention's inventive concept.
Sec. 7(b) provides a "sense of Congress" that recent Supreme Court decisions have harmed progress of science and the useful arts, the Constitutional justification for amending the statute in support of the enumerated Article I power of Congress to establish the patent system. The amendment is direct in its effects on this jurisprudence in Sec. 7(b)(3):
[T]his amendment effectively abrogates Alice Corp. v. CLS Bank International, 134 S. Ct. 2347 (2014) and its predecessors to ensure that life sciences discoveries, computer software, and similar inventions and discoveries are patentable, and that those patents are enforceable.
Sec. 8(a) restores the prior definitions of the prior art, now enumerated rather than designated by subsection of Section 102.
Sec. 8(b) eliminates from the prior art disclosures derived from the inventor, or that occur during the one-year grace period, or are commonly owned or subject to an obligation to assign:
(b) DISCLOSURES IN PATENT APPLICATIONS AND PATENTS.—A disclosure shall not be prior art to a claimed invention under this section if before the issuance of a patent—
(1) the information disclosed was obtained directly or indirectly from the inventor or a joint inventor;
(2) the information disclosed to the Office or another party during the one-year period prior to the date of the application for patent had, before a patent application for the information was effectively filed, been publicly disclosed by the inventor or joint inventor; or
(3) the information disclosed and the claimed invention, not later than the effective filing date of the claimed invention, were owned by the same person or subject to an obligation of assignment to the same person.
Sec. 9(a) "restore[s] patents as a property right," by adding new § 106 to the Patent Act:
§ 106. Private property patent right
A patent right is a private property right secured to an inventor upon issuance of the patent that shall only be revoked by a court ruling in a judicial proceeding, unless the patent owner consents to an administrative or other procedure.
And Sec. 9(c) asserts a "sense of Congress" that "recent jurisprudence of the United States Supreme Court, including Impression Products Inc. v. Lexmark International, Inc., have harmed the progress of science and the useful arts . . ." Unmentioned but equally disturbing to the concept that a patent is a private property right is the Court's decision in SAS Institute Inc. v. Iancu; that decision would have much less relevance if Sections 4 and 9 of this bill were enacted into law. But the bill's specific recitation of Lexmark also indicates that this provision is intended to overturn the post-sale restrictions the Court imposed on patentees in Lexmark, making explicit that the patent property right overcomes the common law principle against restraints on alienating property.
Sec. 10(a) would end "automatic" publication of U.S. patent applications, authorizing publication only upon the request of the patent applicant, and eliminating public disclosure of the file history of any patent application until patent grant.
Sec. 10(b) asserts a "sense of Congress" that:
(1) automatic publication of patent applications after 18 months has harmed the progress of science and the useful arts by creating ''prior art'' by operation of law that prevents a patent owner from applying for a patent on the same invention if a patent does not issue; and
(2) automatic publication of patent applications encourages early disclosure of claimed inventions and subjects innovative inventions and discoveries reduced to practice in the United States to theft or appropriation by foreign competitors.
Sec. 11(a) proposes revised language in 35 U.S.C. § 282 that in addition to the statutory presumption of validity of a patent, each claim must be presumed valid "independently of the validity of other claims" and that a dependent claim must be presumed valid even if dependent on an invalid claim.
Sec. 11(b) adds a patent term tolling provision, in new § 282(d), including a damages provision for validity challenged brought in "bad faith":
TOLLING OF PATENT TERM DURING VALIDITY CHALLENGE.—In an action involving a patent where the validity of the patent has been challenged, the patent term shall be tolled from the time the validity of the patent is challenged to the time of resolution of the validity issue by the court. The patent term shall resume once the validity challenge is resolved. The court may award damages to the patent owner in a case in which another party brought a validity claim against the patent in bad faith.
Sec. 12 amends § 283 to include a mandatory permanent injunction upon a finding of infringement, which can be overcome only by a showing by the infringer by clear and convincing evidence that the patentee would not be irreparably harmed. A finding of irreparable harm does not require the patentee to be making or selling a product covered by the patent. This provision effectively abrogates the Supreme Court's decision in eBay Inc. v. MercExchange, LLC, which introduced into the injunction calculus considerations such as whether the patentee was practicing the invention (which in itself was not supported by the express language of the statute).
Sec. 13 restores the best mode requirement of § 112(a) as a defense in patent infringement litigation.
The bill has been referred to House Judiciary Committee, where its prospects are, to say the least, very uncertain.