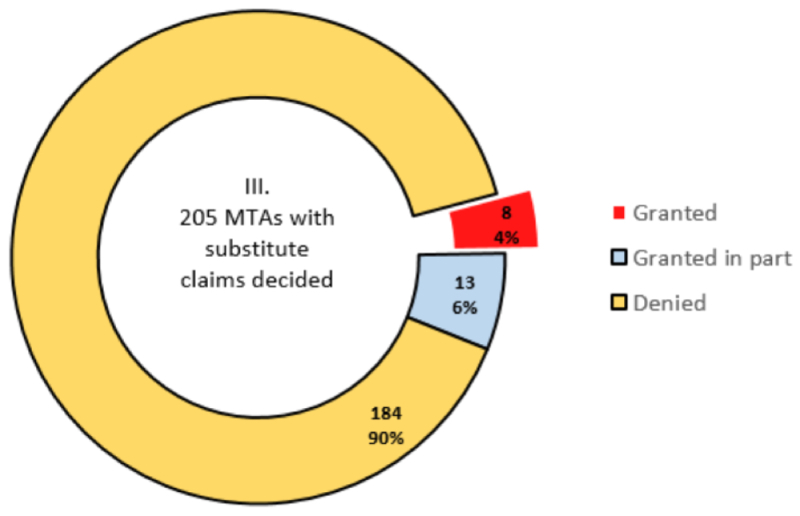
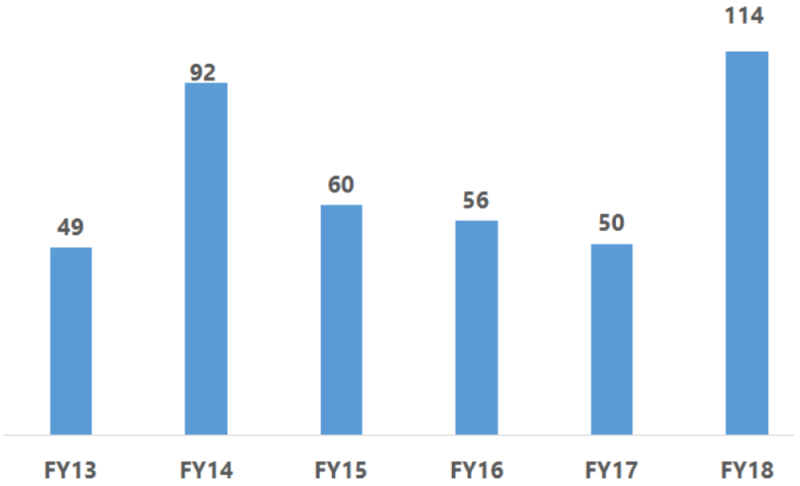
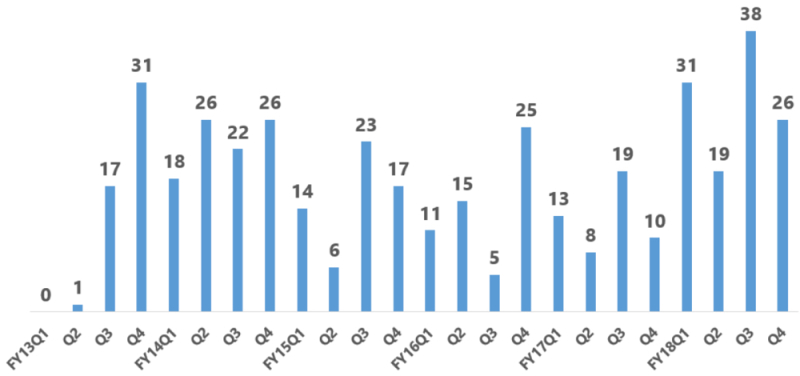
By Kevin E. Noonan —
 The U.S. Patent and Trademark Office today granted U.S. Patent 10,227,611 to Jennifer Doudna, Martin Jinek, Krzysztof Chylinski, and Emmanuelle Charpentier, the patent entitled "Methods and compositions for RNA-directed target DNA modification and for RNA-directed modulation of transcription" and assigned to the Regents of the University of California, the University of Vienna and Emmanuelle Charpentier herself. Claim 1 is representative:
The U.S. Patent and Trademark Office today granted U.S. Patent 10,227,611 to Jennifer Doudna, Martin Jinek, Krzysztof Chylinski, and Emmanuelle Charpentier, the patent entitled "Methods and compositions for RNA-directed target DNA modification and for RNA-directed modulation of transcription" and assigned to the Regents of the University of California, the University of Vienna and Emmanuelle Charpentier herself. Claim 1 is representative:
1. A method of modifying a target DNA molecule in a cell, the method comprising contacting a target DNA molecule inside of a cell with: (a) a Cas9 protein; and (b) a single molecule DNA-targeting RNA comprising, in 5' to 3' order: (i) a targeter-RNA that hybridizes with a target sequence of the target DNA molecule, (ii) a nucleotide linker; and (iii) an activator-RNA that hybridizes with the targeter-RNA to form a double-stranded RNA duplex, wherein (a) forms a complex with (b) and the target DNA molecule is modified.
As it turns out, this is the third patent granted to these inventors; an application allowed several months ago (see "Whither CRISPR? University of California/Berkeley Granted Another CRISPR Patent") has not issued due to formalities deficiencies according to a paper sent to the inventors on February 26th. The earliest patent in this family, U.S. Patent No. 10,000,772, is limited to bacterial cells:
1. A method of modifying a target DNA molecule, the method comprising: contacting a target DNA molecule having a target sequence with a complex comprising: (a) a Cas9 protein; and (b) a DNA-targeting RNA comprising: (i) a targeter-RNA that hybridizes with the target sequence, and (ii) an activator-RNA that hybridizes with the targeter-RNA to form a double-stranded RNA (dsRNA) duplex of a protein-binding segment, wherein the activator-RNA hybridizes with the targeter-RNA to form a total of 10 to 15 base pairs, wherein said contacting takes place outside of a bacterial cell and outside of an archaeal cell, thereby resulting in modification of the target DNA molecule.
The Office's reasoning for allowing the '611 patent parallels that for allowed U.S. Patent Application Publication No. US 2014/0068797 A1, to the extent that these claims were found patentable over prior art because the three RNA species (targeter-RNA, nucleotide linker, and activator-RNA) are covalently linked which does not occur in nature. Claim 165 is representative of the claims allowed but not yet granted in the '797 publication:
165. A method of cleaving a nucleic acid comprising contacting a target DNA molecule having a target sequence with an engineered and/or non-naturally-occurring Type II Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR)-CRISPR associated (Cas) (CRISPR-Cas) system comprising
a) a Cas9 protein; and
b) a single molecule DNA-targeting RNA comprising
i) a targeter-RNA that hybridizes with the target sequence, and
ii) an activator-RNA that hybridizes with the targeter-RNA to form a double-stranded RNA duplex of a protein-binding segment,
wherein the activator-RNA and the targeter-RNA are covalently linked to one another with intervening nucleotides,
wherein the single molecule DNA-targeting RNA forms a complex with the Cas9protein,
whereby the single molecule DNA-targeting RNA targets the target sequence, and the Cas9 protein cleaves the target DNA molecule.
And the Office granted U.S. Patent No. 10,113,167 on October 29, 2018, reciting this representative claim:
1. A non-naturally occurring DNA-targeting RNA, or a nucleic acid encoding the non-naturally occurring DNA-targeting RNA, wherein the non-naturally occurring DNA-targeting RNA comprises: (a) a targeter-RNA comprising: (i) a first nucleotide sequence that is complementary to a target sequence of a target DNA molecule, and (ii) a second nucleotide sequence that hybridizes with an activator-RNA, wherein the first and second nucleotide sequences are heterologous to one another; and (b) the activator-RNA, which hybridizes with the second nucleotide sequence of the targeter-RNA to form a double-stranded RNA (dsRNA) duplex of a protein-binding segment, wherein the activator-RNA hybridizes with the targeter-RNA to form a total of 8 to 15 base pairs, wherein the non-naturally occurring DNA-targeting RNA is capable of forming a complex with a Cas9 polypeptide and targeting the complex to the target sequence of the target DNA molecule.
The Reasons for Allowance in the '611 patent make note of the scope of these claims (all cell types) compared with prior patented methods to the Broad Institute (limited to eukaryotic cells), supporting the apprehension that potential developers of CRISPR technology will need to license California's patents as well as the Broad's to practice CRISPR in eukaryotic cells. This will undoubtedly complicate how the technology develops, as well as making it more expensive and difficult to bring to market.
Several other pending applications have claims in condition for allowance but have had prosecution suspended pending PTAB consideration of whether to declare one or more additional interferences; these include:
• U.S. Application No. 15/435,233, filed on 2-16-2017, which claims the benefit of U.S. Application No. 15/138,604;
• U.S. Application No. 15/925,544, filed on 3-19-2018, which claims the benefit of U.S. Application No. 15/138,604;
• U.S. Application No. 15/947,700, filed on 4-6-2018, which claims the benefit of U.S. Application No. 15/138,604;
• U.S. Application No. 15/947,718, filed on 4-6-2018, which claims the benefit of U.S. Application No. 15/138,604;
• U.S. Application No. 15/981,808, filed on 5-16-2018, which claims the benefit of U.S. Application No. 15/138,604; and
• U.S. Application No. 15/981,809, filed on 5-16-2018, which claims the benefit of U.S. Application No. 15/138,604.
Applicants have petitioned the Office to permit these claims to come to issue on the grounds that there is no interference-in-fact between these claims and the claims of the Broad patent estate, but the Office is insisting on following procedures to permit a PTAB APJ to render a decision in due course.
Finally these applications are in the initial stages of prosecution on the merits:
• U.S. Application No. 16/136,159, filed on 9-19-2018, which claims the benefit of U.S. Application No. 15/138,604 — subject to a restriction requirement issued on February 27th; and
• U.S. Application No. 16/136,165, filed on 9-19-2018, which claims the benefit of U.S. Application No. 15/138,604 — subject to a restriction requirement issued on January 30th.
These pending applications have not published and thus are not available on PAIR:
• 16/201,836 filed on 11-27-2018 claims the benefit of U.S. Application No. 13/842,859
• 16/201,848 filed on 11-27-2018 claims the benefit of U.S. Application No. 13/842,859
• 16/201,853 filed on 11-27-2018 claims the benefit of U.S. Application No. 13/842,859
• 16/201,855 filed on 11-27-2018 claims the benefit of U.S. Application No. 13/842,859
• 16/201,862 filed on 11-27-2018 claims the benefit of U.S. Application No. 13/842,859
• 16/201,865 filed on 11-27-2018 claims the benefit of U.S. Application No. 13/842,859
• 16/276,343 filed on 02-14-2019 claims the benefit of U.S. Application No. 13/842,859
• 16/276,348 filed on 02-14-2019 claims the benefit of U.S. Application No. 13/842,859
• 16/276,352 filed on 02-14-2019 claims the benefit of U.S. Application No. 13/842,859
• 16/276,356 filed on 02-14-2019 claims the benefit of U.S. Application No. 13/842,859
• 16/276,361 filed on 02-14-2019 claims the benefit of U.S. Application No. 13/842,859
• 16/276,365 filed on 02-14-2019 claims the benefit of U.S. Application No. 13/842,859
• 16/276,368 filed on 02-14-2019 claims the benefit of U.S. Application No. 13/842,859
• 16/276,374 filed on 02-14-2019 claims the benefit of U.S. Application No. 13/842,859 and
• 16/277,090 filed on 02-15-2019 claims the benefit of U.S. Application No. 13/842,859