By Kevin E. Noonan —
Durum wheat, Triticum turgidum L. ssp. durum (Desf.) Husn., used principally for pasta production, was derived from wild emmer wheat, T. turgidum ssp. dicoccoides (Körn. ex Asch. & Graebn.) Thell. from domesticated emmer wheat, T. turgidum ssp. dicoccum (Schrank ex Schübl.) Thell. about 10,000 years ago in the Fertile Crescent. Domesticated wheat became established as a human food source 1,500-2,000 years ago, characterized by an initial domestication phase followed by continued evolution through further domestication and selective breeding.
This week, a group* of international researchers disclosed the genome DNA sequence of domesticate Durum wheat in a paper entitled "Durum wheat genome highlights past domestication signatures and future improvement targets" and published in Nature Genetics. The paper reports elucidation of the 10.45 Gigabase genome from durum wheat cultivar Svevo and identified regions of the wheat genome selected for by thousands of years of selective breeding by humans. For example, a region on chromosome 5B carries a locus of a metal transporter (TdHMA3-B1) having a non-functional variant associated with cadmium accumulation and characteristic of Durum cultivars and not found in wild emmer wheat. The durum wheat genome is illustrated in the paper as follows:
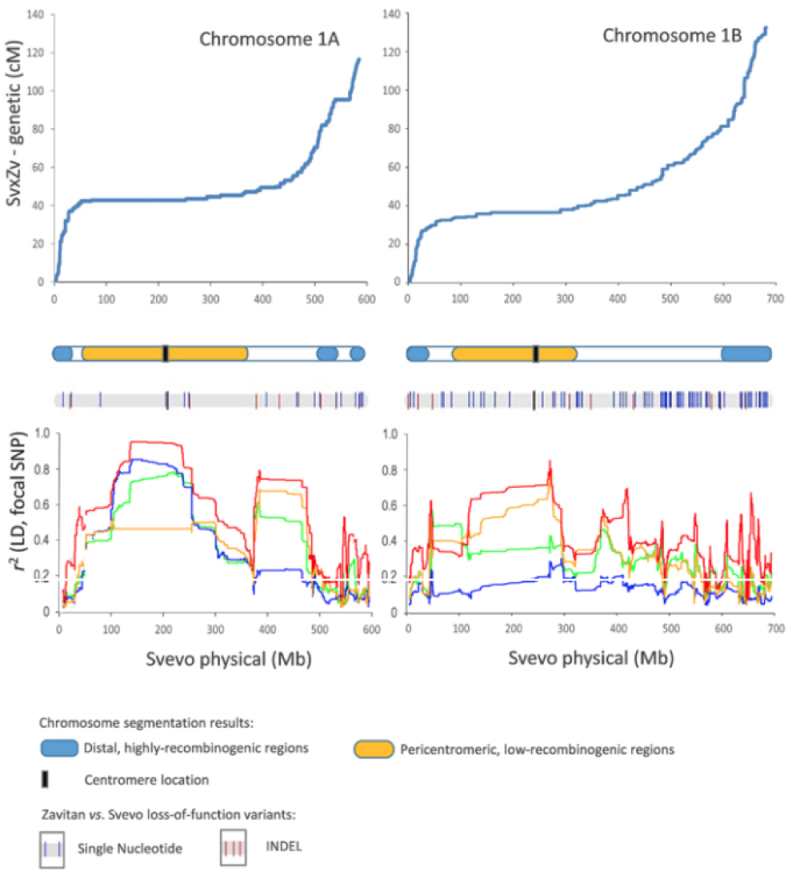
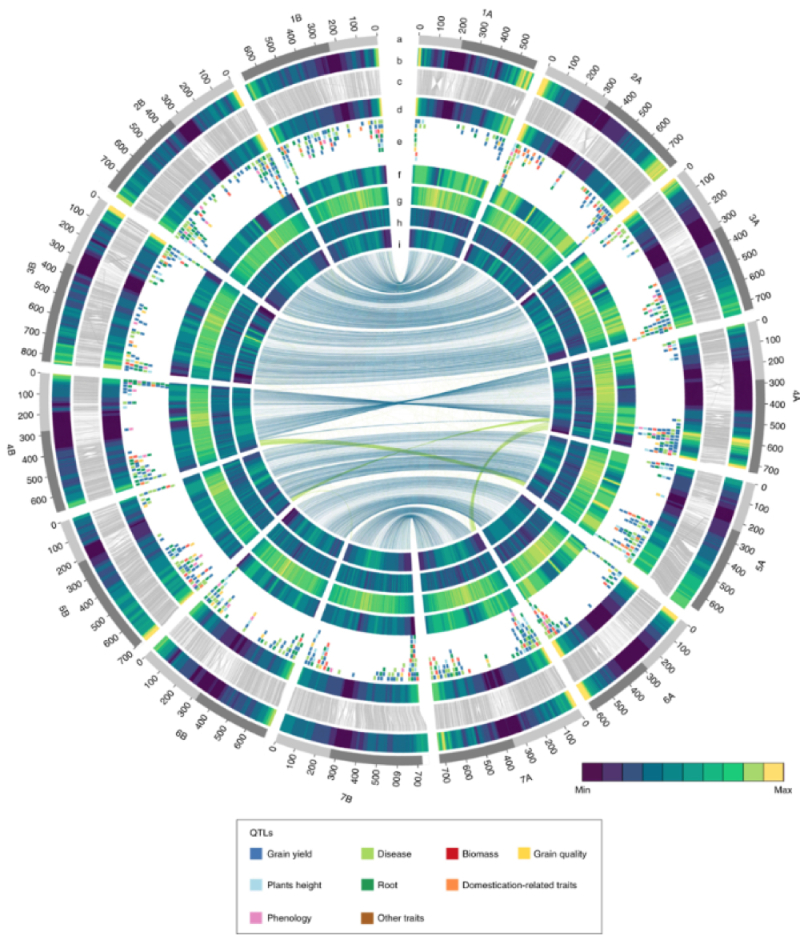 The genome is segregated into 14 chromosomes, designated 1A/1B through 7A/7B in the Figure. The distal regions of these chromosomes, representing about 22% of the genome, are highly recombinogenic according to the authors, as illustrated by high-density single nucleotide polymorphism (SNP) maps, showing an almost linear relationship between physical and genetic distance as ascertained by recombination. The pericentromeric regions, on the other hand, show almost no recombination and represent about 44% of the genome. Analysis disclosed 66,559 "high confidence (HC)" genes, with about 91% of these being detected as expressed in assembled gene expression data sets; these relationships are illustrated in Supplementary Figure 1 from the paper:
The genome is segregated into 14 chromosomes, designated 1A/1B through 7A/7B in the Figure. The distal regions of these chromosomes, representing about 22% of the genome, are highly recombinogenic according to the authors, as illustrated by high-density single nucleotide polymorphism (SNP) maps, showing an almost linear relationship between physical and genetic distance as ascertained by recombination. The pericentromeric regions, on the other hand, show almost no recombination and represent about 44% of the genome. Analysis disclosed 66,559 "high confidence (HC)" genes, with about 91% of these being detected as expressed in assembled gene expression data sets; these relationships are illustrated in Supplementary Figure 1 from the paper:
 The periocentric regions also showed a diminution in diversity that arose during domestication from wild emmer wheat to domesticated durum emmer wheat, particularly associated with chromosomes 2A, 4A, 4B, 5A, 5B, 6A, and 6B. As disclosed in the paper, "several reductions in diversity (75 with diversity reduction index > 2) were specifically associated with breeding of modern durum cultivars, including some associated with disease resistance (for example, Sr13; ref. 29 and Lr14; ref. 30) and grain yellow pigment content loci (for example, Psy-B1; ref. 31)."
The periocentric regions also showed a diminution in diversity that arose during domestication from wild emmer wheat to domesticated durum emmer wheat, particularly associated with chromosomes 2A, 4A, 4B, 5A, 5B, 6A, and 6B. As disclosed in the paper, "several reductions in diversity (75 with diversity reduction index > 2) were specifically associated with breeding of modern durum cultivars, including some associated with disease resistance (for example, Sr13; ref. 29 and Lr14; ref. 30) and grain yellow pigment content loci (for example, Psy-B1; ref. 31)."
Comparison between the Durum wheat genome and the progenitor wild emmer wheat genome showed strong overall synteny and high degree of similarity in total HC gene number (66,559 vs. 67,182). LTR-Retrotransposon insertion sites were also found to be syntenic, resulting from the relatively short time since divergence of these cultivars. Analysis of related gene clustering showed 36,434 "unigene" groups, 28,794 of which (79%) belonged to clusters having at least two members and 21% (7,640) being single genes.
In particular, the paper describes a candidate gene, Cdu-B1, located on chromosome 5B associated with differences in cadmium (Cd) accumulation in durum wheat. High throughput sequencing and comparison between genomic regions associated with this gene identified a region of increased sequence variation that was determined to encode a metal transporter, TdHMA3-B1; these experiments are illustrated in Supplementary Figure 6:

Alleles of the TdHMA3-B1 gene were found to discriminate between low- and high-Cd accumulating wheat genotypes, as illustrated in Supplementary Figure 7:
 The extensive genetic comparisons (set forth predominantly in the Supplementary datasets) provided the bases for the authors' conclusions:
The extensive genetic comparisons (set forth predominantly in the Supplementary datasets) provided the bases for the authors' conclusions:
Access to the fully annotated genome sequence in combination with the wealth of genotypic, genetic mapping and gene expression data provides great potential for future innovation for the wheat scientific community and the breeding sector. Gene discovery, quantitative trait loci (QTL) cloning and the precision of genomics-assisted breeding to enhance grain quality and quantity of pasta wheat will benefit from the resources presented here. Furthermore, the durum sequence provides a fundamental tool to more effectively bridge and harness the allelic diversity present in wheat ancestors most of which remains largely untapped.
* Marco Maccaferri, Danara Ormanbekova, Elisabetta Frascaroli, Simona Corneti, Silvio Salvi & Roberto Tuberosa, Department of Agricultural and Food Sciences, University of Bologna, Bologna, Italy; Marco Maccaferri, Pasquale De Vita, Daniela Marone, Nicola Pecchioni & Anna M. Mastrangelo, Research Centre for Cereal and Industrial Crops, Foggia, Italy; Neil S. Harris, Kevin Y. H. Liang & Gregory J. Taylor, Department of Biological Sciences, University of Alberta, Edmonton, Alberta, Canada; Sven O. Twardziok, Heidrun Gundlach, Manuel Spannagl, Danara Ormanbekova, Thomas Lux, Verena M. Prade & Klaus F. X. Mayer, Helmholtz Zentrum München, Plant Genome and Systems Biology, Neuherberg, Germany; Raj K. Pasam, Reem Joukhadar & Matthew J. Hayden, Agriculture Victoria, Agribio Centre for AgriBioscience, Bundoora, Victoria, Australia; Sara G. Milner, Axel Himmelbach, Martin Mascher & Nils Stein, Leibniz Institute of Plant Genetics and Crop Plant Research, Gatersleben, Germany; Martin Mascher, German Centre for Integrative Biodiversity Research Halle-Jena-Leipzig, Leipzig, Germany; Paolo Bagnaresi, Primetta Faccioli, Francesca Desiderio, Caterina Marè, Cristina Crosatti, Erica Mica, Elisabetta Mazzucotelli & Luigi Cattivelli, Research Centre for Genomics and Bioinformatics, Fiorenzuola d'Arda, Italy; Paolo Cozzi, Massimiliano Lauria, Barbara Lazzari, Alessandra Stella & Aldo Ceriotti, National Research Council—Institute of Agricultural Biology and Biotechnology, Milano, Italy; Andrea Manconi, Matteo Gnocchi, Marco Moscatelli & Luciano Milanesi, National Research Council—Institute of Biomedical Technologies, Segrate, Italy; Raz Avni, Jasline Deek & Assaf Distelfeld, School of Plant Sciences and Food Security, Tel Aviv University, Tel Aviv, Israel; Sezgi Biyiklioglu & Hikmet Budak, Montana State University, Bozeman, MT, USA; Gabriella Sonnante, National Research Council—Institute of Biosciences and Bioresources, Bari, Italy; Hakan Özkan, Çukurova University, Faculty of Agriculture, Department of Field Crops, Adana, Turkey; Benjamin Kilian, Global Crop Diversity Trust, Bonn, Germany; Reem Joukhadar, Department of Animal, Plant and Soil Sciences, La Trobe University, Bundoora, Victoria, Australia; Domenica Nigro, Department of Soil, Plant and Food Sciences, University of Bari Aldo Moro, Bari, Italy; Agata Gadaleta, Department of Agricultural and Environmental Science, University of Bari Aldo Moro, Bari, Italy; Shiaoman Chao, Justin D. Faris & Steven S. Xu, United States Department of Agriculture, Agricultural Research Service, Edward T. Schafer Agricultural Research Center, Fargo, ND, USA; Arthur T. O. Melo & Iago Hale, Department of Agriculture, Nutrition, and Food Systems, University of New Hampshire, Durham, NH, USA; Mike Pumphrey, Department of Crop and Soil Sciences, Washington State University, Pullman, WA, USA; Krystalee Wiebe, Jennifer Ens, Ron P. MacLachlan, John M. Clarke, Sean Walkowiak & Curtis J. Pozniak, Crop Development Centre and Department of Plant Sciences, University of Saskatchewan, Saskatoon, Saskatchewan, Canada; Andrew G. Sharpe & Chu Shin Koh, Global Institute for Food Security, University of Saskatchewan, Saskatoon, Saskatchewan, Canada; Ron Knox, Swift Current Research and Development Centre, Agriculture and Agri-Food Canada, Swift Current, Saskatchewan, Canada; Anna M. Mastrangelo, Research Centre for Cereal and Industrial Crops, Bergamo, Italy; Matthew J. Hayden, School of Applied Systems Biology, La Trobe University, Bundoora, Victoria, Australia; and Klaus F. X. Mayer, School of Life Sciences Weihenstephan, Technical University Munich, Freising, Germany
 The Intellectual Property Owners Association (IPO) will offer a one-hour webinar entitled "IPR Estoppel One Year After SAS" on April 25, 2019 from 2:00 to 3:00 pm (ET). Grantland Drutchas of McDonnell Boehnen Hulbert & Berghoff LLP; James Hietala of Intellectual Ventures Management, LLC; and Eliot Williams of Baker Botts LLP will address how district courts have applied IPR estoppel in the year since SAS v. Iancu was decided, and will not only highlight recent cases, but will also provide tactics for both plaintiffs and defendants to make arguments about the proper scope of IPR estoppel in district court litigation. The panel will address the following topics:
The Intellectual Property Owners Association (IPO) will offer a one-hour webinar entitled "IPR Estoppel One Year After SAS" on April 25, 2019 from 2:00 to 3:00 pm (ET). Grantland Drutchas of McDonnell Boehnen Hulbert & Berghoff LLP; James Hietala of Intellectual Ventures Management, LLC; and Eliot Williams of Baker Botts LLP will address how district courts have applied IPR estoppel in the year since SAS v. Iancu was decided, and will not only highlight recent cases, but will also provide tactics for both plaintiffs and defendants to make arguments about the proper scope of IPR estoppel in district court litigation. The panel will address the following topics:











