By Antony Craggs —
Since the introduction of the doctrine of equivalents in the UK in Actavis v Lilly, it has been questioned whether a claim can be extended by said doctrine (so that a product infringes the claim) where such an extension would render the claim obvious over the prior art at the priority date. In Technetix v Teleste, HHJ Hacon has offered obiter guidance on the point.
Background — Actavis v Lilly
Actavis v Lilly introduced the following questions when assessing infringement:
1. Does the variant infringe any of the claims as a matter of normal interpretation; and, if not,
2. Does the variant nonetheless infringe because it varies from the invention in a way or ways which is or are immaterial?
The latter question is further addressed by the following three questions:
1. Notwithstanding that it is not within the literal meaning of the relevant claim(s) of the patent, does the variant achieve substantially the same result in substantially the same way as the invention, i.e. the inventive concept revealed by the patent?
2. Would it be obvious to the person skilled in the art, reading the patent at the priority date, but knowing that the variant achieves substantially the same result as the invention, that it does so in substantially the same way as the invention?
3. Would such a reader of the patent have concluded that the patentee nonetheless intended that strict compliance with the literal meaning of the relevant claim(s) of the patent was an essential requirement of the invention?
In doing so, the approach to construction of the claims of patent (namely, purposive) remained the same but the scope of protection was broadened. As validity was not in issue in the case, however, the nexus between validity and the doctrine of equivalents was not addressed. This was a particularly acute issue because English law, prior to Actavis v Lilly, conflated the test of novelty and infringement, namely a claim lacked novelty if the prior publication disclosed subject-matter which, if performed, would necessarily infringe the claim. In essence, the test for novelty elided construction with infringement, with the latter now including the doctrine of equivalents.
Generics v Teva
This issue was considered at first instance by Mr Justice Arnold in Generics v Teva. He held that it was no longer the law that a claim lacked novelty if the prior publication disclosed subject-matter which, if performed, would necessarily infringe the claim. Rather, the claim would only lack novelty if the prior publication disclosed subject-matter which fell within the claim in its proper construction. It was not sufficient that the subject-matter would infringe the claim applying the doctrine of equivalents. We await a more senior court to consider the issue.
This leaves a potential lacuna between validity and infringement. This is best demonstrated as follows:
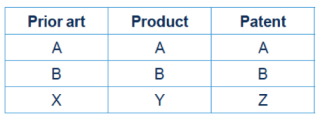
Prior to Actavis v Lilly, if a patent's features were novel and inventive (feature Z over X), a patent was valid.
Further, if a patent's features were not embodied in the alleged infringement (Z is not Y), a patent was not infringed. It followed that under the above example, the patent would be valid but not infringed.
The position potentially changes after Actavis v Lilly because the patent can now be infringed by a product which has an equivalent feature (Y is the equivalent of Z). If Y is obvious over X (but Z is not obvious over X), the patent remains valid but is infringed by an "obvious" product. This is an untenable position: it is a fundamental principle of patent law that a party must be free to do that which was either not new or obvious at the priority date.
Gillette defence
A Gillette defence may be the answer. It is the latter half of the last sentence which is important. This is because, in practice, a Gillette defence has been applied as a counterclaim challenging the validity of the patent, not as a stand-alone defence. The rationale for this was two-fold:
1. absent the doctrine of equivalents, a patent could not be valid and infringe a product which was obvious over the prior art (there was, therefore, no need for a stand-alone defence); and
2. as a matter of public policy it was appropriate to address the validity of the patent (as a right in rem), rather than as a defence (as a right in personam.
So is a new stand-alone defence needed?
Technetix v Teleste
In the case at hand, Technetix brought a claim against Teleste for infringement of its patent, GB 2,383,473 for a cable tap unit for receiving and delivering a cable television or Internet signal to subscribers.
Teleste counterclaimed, challenging the validity of the patent.
Following an adjournment of the trial (see Technetix v Teleste: Adjournment of Patent Trial), the claim was heard before the Intellectual Property Enterprise Court (IPEC) in November 2018. The patent was held to be invalid, but the court provided its obiter comments on infringement. This defence was fully pleaded on both sides and argued at the trial.
HHJ Hacon declined to hold whether such a defence exists. However, he offered a proposal as to how such a defence (if it exists) may operate. Drawing on the Germany Federal Supreme Court in Formstein, he posited that a defence could be adopted in English law so that, if an accused product or process is an equivalent and for that reason is nominally within the scope of the claim, but the equivalent would have lacked novelty or inventive step over the prior art at the priority date, then it is deemed to fall outside the scope of the claim.
HHJ Hacon noted that such a defence also exists under Dutch law, see Core Distribution Inc v Lidl Nederland GmbH, and was analogous to the principle of ensnarement in the USA.
Applying this to the facts (on the assumption that the patent was valid), HHJ Hacon held that the patent was not infringed on a purposive construction, but would be under the doctrine of equivalents. Assuming a Formstein defence existed, he concluded that the defendant was entitled to the defence.
* Mr. Craggs is a partner and solicitor with D Young & Co
This article was reprinted with permission from D Young & Co.
 June 12-14, 2019 – Patent Fundamentals Bootcamp 2019: An Introduction to Patent Drafting, Prosecution, and Litigation (Practising Law Institute) – Chicago, IL
June 12-14, 2019 – Patent Fundamentals Bootcamp 2019: An Introduction to Patent Drafting, Prosecution, and Litigation (Practising Law Institute) – Chicago, IL