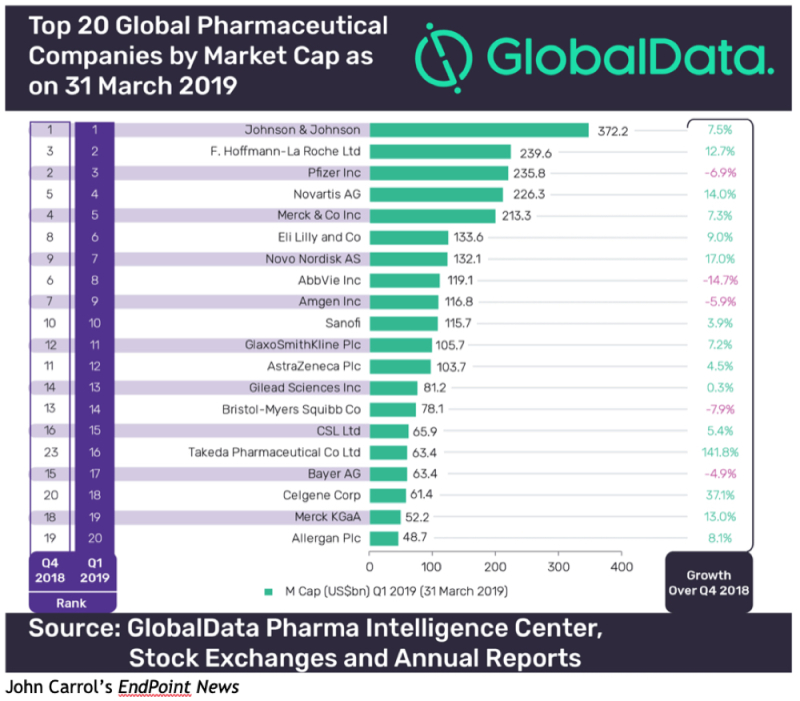
By Kevin E. Noonan —
 On October 18th, Junior Party (the University of California, Berkeley; the University of Vienna; and Emmanuelle Charpentier (collectively, "CVC") filed its authorized opposition to Substantive Motion No. 1 from Senior Party the Broad Institute (and its partners as Senior Party, Harvard University and MIT), which asked for judgment in Interference No. 106,115 on the basis that CVC was estopped by prior judgment of no interference in fact in Interference No. 106,048 between these parties.
On October 18th, Junior Party (the University of California, Berkeley; the University of Vienna; and Emmanuelle Charpentier (collectively, "CVC") filed its authorized opposition to Substantive Motion No. 1 from Senior Party the Broad Institute (and its partners as Senior Party, Harvard University and MIT), which asked for judgment in Interference No. 106,115 on the basis that CVC was estopped by prior judgment of no interference in fact in Interference No. 106,048 between these parties.
To recap, the Broad's motion asserted these arguments in favor of judgment:
CVC is estopped from getting a "second bite at the apple" in this interference by the provisions of 37 C.F.R. § 41.127(a)(1) and MPEP § 2308.03(b) (interference estoppel) because:
- this interference is directed to the same subject matter as the previous '048 patent (using the inclusion of the same Broad patents in this interference and the correspondence between the count in the '048 interference and claim 1 in involve U.S. Patent No. 8,697,359 in support)
- CVC is estopped from pursuing this interference on the grounds of judgment estoppel regarding issues that were raised or could have been raised in the earlier interference:
Judgment. (a) Effect within Office—(1) Estoppel. A judgment disposes of all issues that were, or by motion could have properly been, raised and decided. A losing party who could have properly moved for relief on an issue, but did not so move, may not take action in the Office after the judgment that is inconsistent with that party's failure to move, except that a losing party shall not be estopped with respect to any contested subject matter for which that party was awarded a favorable judgment [emphasis added].
The Broad argued that CVC had the opportunity in the '048 interference to file a responsive motion to the Broad's motion of no interference-in-fact to add claims directed to eukaryotic applications of CRISPR technology but did not. The Broad argues that this was a strategic decision by CVC to have only its "environment-free" claims in the interference. Furthermore, the Broad contends that it was intentional, citing to CVC's colloquy before the Board when the parties discussed which motion the Board would authorize, where CVC expressly "reserved" the ability to file such responsive motions.
The Broad's motion also asserts that the Board should not permit CVC to maintain the "cloud of uncertainty" regarding its patents, in its public statements and by pursuing "serial" interferences against the same Broad patents.
As is required under the procedural rules in interferences, CVC's opposition addresses each of these contentions in turn, including any disagreement over the facts the Broad put forward in support. Thus, CVC's first asserted opposition argument was that "judgment estoppel" (quotation marks in opposition brief) does not exist in any form that would prelude this interference. CVC's counter to the Broad's argument focuses on the existence vel non of a "losing" party in cases where no interference-in-fact are found. The brief characterizes as false the assumption that the subject matter of this interference is the same as the subject matter of the '048 interference. In the prior interference, it is undisputed that the Broad's claims were limited to practicing CRISPR in eukaryotic cells while CVC's claims-in-interference had no such restriction. That restriction (embodied in the Count) was imposed by the Board when the '048 interference was declared (and the Board never ruled on CVC's motion to change the count to remove this restriction. The scope of that earlier interference is different from this one, which is frankly directed towards using CRISPR solely in the eukaryotic background. According to CVC, this is the first time the priority question on practicing CRISPR solely in the eukaryotic cell context has been before the Board, and thus there should be no estoppel.
With regard to the Broad's arguments related to Rule 127, CVC contends that the Broad has misconstrued the meaning of the first sentence of the Rule. There is no per se "failure to move" requirement devoid of the need for a losing party in the prior interference (which does not exist in this case). The brief takes the opportunity to assert its priority argument in this section, arguing that CVC was "the first party to file a patent application identifying the necessary and sufficient components of a CRISPR-Cas system that cleaves or edits DNA," starting with its May 25, 2012 filing date, importantly encompassing its use without regard to cellular milieu.
The brief further addresses the assertions made that CVC was executing a "tactical decision" to "wage [a] serial interference campaign." On the contrary, CVC argues that it had suggested a count in the '048 interference that was cellular milieu-free, and it was the Board that restricted the count to use in eukaryotic cells. Nor, CVC contends, can its efforts to obtain patents on systems and uses of CRISPR in eukaryotic cells as "a mere attack" on the Broad's claims. CVC is jut doing what it was always intending to do, obtain broad and specific claim coverage for uses of CRISPR broadly. CVC also reminds the Board that, unlike in the '048 interference in this interference the Board declared it sua sponte (further blunting any allegations of manipulation or scheming, according to CVC). Having found interfering subject matter CVC argues that the Board ha a responsibility to declare an interference to resolve the priority issue under 35 U.S.C. §§ 102(g) and 135. CVC further argues that it is the Broad that has sought to avoid Board determination of the priority question, and points to its alleged motivation to do so: "[the] Broad was actually the fourth entity to file a patent application directed to CRISPR in eukaryotes" (citing facts CVC filed in support of tis opposition). The brief also makes sub silentio an argument regarding the Broad's (perfectly proper) prosecution strategy that accelerated examination of its CRISPR patents, and by "changing its applications' designation from post-AIA to pre-AIA during prosecution so that it could swear behind CVC's earlier-filed application." And CVC turns the Broad's victory in the earlier '048 interference against it, characterizing the Broad's successful pursuit of a finding of no interference-in-fact as another way to avoid final Board determination of the priority question (and similarly turns the Broad's allegations that CVC had a responsibility to move that the Board include claims to eukaryotic cell embodiments of CRISPR against the Broad, by alleging that it was the Broad that "could and should have moved to add a eukaryotic claim to CVC's involved application" as the proper alternative to seeking a finding of no interference-in-fact). Reminding the Board of the purported advantages of its alleged strategy, the brief states that:
Broad has enjoyed the benefits of its issued patents for five-and-a- half years but must now finally face the unresolved question of priority of invention for CRISPR in eukaryotes; no principles of collateral estoppel or res judicata prohibit the PTAB from resolving the issue now via this interference. Broad and the public have long been on notice that this issue remained unresolved as between Broad and CVC. The present interference, mandated by §§ 102(g) and 135, presents the proper vehicle for finally resolving priority of invention.
CVC's brief also addresses the estoppel issue, stating that interpreting estoppel as the Broad seeks in its motion would "invite the Office to commit legal error by exceeding its authority." Interference estoppel comes in two flavors, according to the brief: those set forth expressly in the enabling statute and those arising from common law. The latter are impliedly present in the statute because "Congress may be presumed, when enacting a statute granting to an agency adjudicatory authority, to mandate adherence to the doctrine of collateral estoppel," citing Duvall v. Atty. Gen., 436 F.3d 382, 387 (3d Cir. 2006). Here, the statute does not affirmatively set forth an estoppel and thus, according to CVC, any estoppel (including the "judicial estoppel" that is the basis for the Broad's prayer for relief in its motion, must arise from these common law principles. In addition, CVC argues that once Congress gives an agency authority to act, it "cannot promulgate or apply procedural rules in a manner that unilaterally contracts its jurisdiction to make it more narrow than what Congress has provided," citing Union Pac. R. Co. v. Bhd. 13 of Locomotive Engineers & Trainmen, 130 S. Ct. 584, 590 (2009). These arguments are relevant to CVC brief in two ways: first, the PTAB is not capable of promulgating rules on estoppel that go beyond the common law (absent express statutory authority absent here). Nor can the PTAB fail to satisfy its statutory mandate (here) to make priority determinations when priority of invention is in conflict.
With regard to the proper scope of estoppel, CVC argues that the Broad's interpretation of Rule 127 is contrary to these common law estoppel principles. CVC's first argument in support of its opposition in this regard is that this interference does not involve the same subject matter as the prior, '048 interference (making arguments substantially as set forth above on this point). While it is true that the Broad's claims in the earlier interference were limited to eukaryotic cell applications of CRISPR, CVC's were not, and thus this interference considering eukaryotic cell claims of both parties defines different subject matter. CVC's claims limited to eukaryotic cells in this interference are (and must be) patentably distinct from its claims in the '048 interference, because after all that distinction was the basis for the Broad having the PTAB (and the Federal Circuit) decide there was no interference-in-fact in the '048 interference:
The PTAB is now poised to answer who was first to invent CRISPR in eukaryotes. That question was not (and could not have been) litigated or decided in the previous interference given the no interference-in-fact determination. No principle of collateral estoppel or res judicata prevents deciding an issue that was not previously litigated,
according to CVC’s brief, citing B&B Hardware, Inc. v. Hargis Indus., Inc., 135 S. Ct. 1293, 1303 (2015); Biogen MA, Inc. v. Japanese Found. for Cancer Research, 785 F.3d 648, 658 (Fed. Cir. 2015); Purdue Pharma L.P., et al. v. Iancu, 2019 U.S. App. LEXIS 11205 (Fed. Cir. 2019); and Cromwell v. County of Sac, 94 U.S. 351, 353 (1877).
The brief makes a distinction between termination of an interference because no interference-in-fact is found (which, according to CVC, is a dismissal for lack of jurisdiction), which is not the same as a judgment and "does not preclude a second action based on the same cause of action that includes claims that overcome the initial defect of jurisdiction," citing, inter alia, Hughes v. U.S., 71 U.S. 232, 237 (1866). The proper analogy for a finding of no interference-in-fact is dismissal of a district court action for failure to state a claim, where res judicata arises only where the defect in the pleadings has not been cured.
CVC further faults the Broad's estoppel argument by noting that according to its terms Rule 127 raises an estoppel in the context of provoking an interference, whereas the '115 interference was declared sua sponte by the Board (exactly the type of procedural argument that frequently resonates with the Board; see "Sigma-Aldrich Tried Again"). Similarly, CVC's brief distinguishes MPEP 2308.03(b) from MPEP 2308.03(c) on the basis that the former rule of practice bars "any further interference between the same 2 parties for claims to the same invention as the count of the interference, [and] not any subsequent interference that presents a new question not previously litigated" (emphasis in brief). (And even if the PTAB disagrees with how CVC has parsed the MPEP, the brief asserts that "[t]o the extent that MPEP 2308.03(b) is construed to bar the present interference, the MPEP is contrary to the law and therefore invalid.") Finally, on these matters CVC disagrees that the scope of the count of the '048 interference has any weight or relevance, because if there was no interference-in-fact in the '048 interference there was no count that properly defined interfering subject matter.
CVC's brief then turns to the Broad's argument that CVC was under an obligation to move to include claims in the '048 interference directed to eukaryotic embodiments of CRISPR and that failing to do so raises an estoppel. Here CVC again raises objection to the Broad's interpretation of Rule 127, asserting (again) that the Rule requires there to be a losing party for estoppel to arise and a finding of no interference-in-fact does not produce a losing party. (The brief cites voluminous authority regarding the proper relationship between clauses or provisions of rules or statutes and their interpretation in support of this argument.) The brief notes (at least in part in contrast) that the Broad cited no authority for its (mis)interpretation of the Rule, and that its citation of commentary on the rulemaking that resulted in the provisions of the MPEP cited in the Broad's brief either does not support the Broad's interpretation of the Rule nor is the Board bound by them. CVC's conclusion is that:
[N]either Rule 127's text nor the rule-making comments state or imply that, following a judgment of no interference-in-fact, a non-losing party "who could have properly moved for relief on an issue, but did not so move, may not take action in the Office after the judgment that is inconsistent with the party's failure to move." 37 C.F.R. § 41.127(a)(1). That kind of failure-to-move estoppel is expressly reserved for a losing party, and limited to issues for which the losing party was not awarded favorable judgment. Id. Applying estoppel any more broadly would impose an inequitable result on a party that had not lost the prior proceeding.
CVC further blunts (or attempts to do so) in this portion of the brief the Broad's allegations that CVC's actions or inaction in the prior '048 interference were somehow improper or merely strategic, calling these characterizations a "mischaracterize[ation of] the facts." At least one set of these facts set forth in CVC's brief is that the Broad accuses CVC of not moving to include eukaryotic cell CRISPR claims in the '048 interference, when CVC had no such claims in condition for allowance (a requirement for the PTAB to make a priority determination) at that time. CVC also cites a Board Order in the prior interference, which stated that "[u]nder the facts and circumstances of this interference, where [CVC] believes all of its current claims interfere with all of Broad's claims, there is no reason why [CVC] should need to add a new claim. If [CVC]'s claims in other applications are ultimately found to be allowable, [CVC] may suggest additional interferences to the examiner" (emphasis in brief). Clearly, the position argued by the Broad is contrary to the terms of the Board's prior Order (at least as CVC interprets it) and may provide a basis for the Board to disregard this portion of the Broad's argument at a minimum and perhaps deny Substantive Motion No. 1.
The final Sections of CVC's brief address the Broad's assertion of equitable estoppel (which, according to CVC, would result in the Board to "abrogate its statutory mandates to decide interferences under § 135 and to issue patents to all those entitled to them under § 102); certain "policy considerations" that support the PTAB deciding the priority question ("it would contradict the PTAB's legislative mandate if it were to refrain from deciding the outstanding priority of invention issue, particularly where Broad's patents are facially invalid in view of CVC's earlier filed patent applications disclosing CRISPR in eukaryotes"); and that the Board must resolve certain disputed factual issues before deciding to grant the Broad's Substantive Motion No. 1.
Final Section VI of the brief provides CVC with an outcome that could snatch (ultimate) victory from the jaws of a defeat should the Board grant the Broad's Substantive Motion No. 1. Under those circumstances, CVC argues that the PTO has determined that CVC's claims are in condition for allowance but for the priority issues that have arisen under 35 U.S.C. §102(g). Thus, should the Board grant the Broad's motion the impediments to patentability raised by the Examiner would be overcome (i.e., there would be no violation of § 102(g)) and accordingly the Office should issue CVC's patents in due course. This would then give CVC the right to file an action in district court under § 291 to determine which of CVC and the Broad should properly have priority to the subject matter of eukaryotic cell embodiments of CRISPR. While not exactly a poison pill, such an outcome would not necessarily be estopped, because there would be two parties having granted claims to eukaryotic embodiments of CRISPR, and the public interest at a minimum would favor final resolution of the priority question. Of course the Broad recognized the possibility of this outcome and has asserted § 101 as a basis to preclude the Office from granting CVC's patents. While § 101 has been put to a great many uses during the past ten years, it clearly does not apply here (as CVC asserts), because "§ 101 simply bars issuing two patents to the same inventive entity; it says nothing about issuing a patent to a different inventive entity" (which is the provenance of § 102(g)).
The Broad's Reply brief is due March 20, 2020 unless the Board specifies an earlier date (which is likely).