
Patent Law Weblog
-
By Kevin E. Noonan —
 Last year, Judge Illston gifted patentees with a proverbial lump of coal with her decision in Illumina, Inc. v. Ariosa Diagnostics, Inc. by invalidating on summary judgment claims directed to methods for isolating cell-free fetal DNA from maternal DNA on the grounds that they are not patent-eligible under 35 U.S.C. § 101. On December 19, Rep. Danny K. Davis (IL-7), joined by Congressman Paul A. Gosar (AZ-04), introduced a bill that is more like a Christmas gift for inventors, entitled the Inventor Rights Act (H.R. 5478).
Last year, Judge Illston gifted patentees with a proverbial lump of coal with her decision in Illumina, Inc. v. Ariosa Diagnostics, Inc. by invalidating on summary judgment claims directed to methods for isolating cell-free fetal DNA from maternal DNA on the grounds that they are not patent-eligible under 35 U.S.C. § 101. On December 19, Rep. Danny K. Davis (IL-7), joined by Congressman Paul A. Gosar (AZ-04), introduced a bill that is more like a Christmas gift for inventors, entitled the Inventor Rights Act (H.R. 5478).After preliminary sections extolling the contribution of inventors to American innovation and economic advancement, and noting the negative effects on inventor rights by Supreme Court cases including eBay Inc. v. MercExchange, LLC, TC Heartland LLC v. Kraft Foods Group Brands LLC, and PTAB proceedings under the provisions of the Leahy-Smith America Invents Act (AIA), and calling out the practice of "efficient infringement," the bill proposes amending Section 100 of the Patent Act to include a new section (k):
(k) The term 'inventor-owned patent' means a patent with respect to which the inventor of the invention claimed by the patent or an entity controlled by that inventor—
(1) is the patentee; and
(2) holds all substantial rights.For such "inventor-owned patents," the bill proposes a new Section 330, entitled Inventor protections, which include:
(a) Not subjecting such patents to reexamination or review [presumably including post-grant review, inter partes review, or covered business method patent review], or [more broadly], to otherwise make a determination about the validity of an inventor-owned patent" without consent of the inventor-owner;
(b) permitting venue either:
(1) in accordance with section 1400(b) of title 28;
(2) where the defendant has agreed or consented to be sued in the instant action;
(3) where an inventor named on the patent in suit conducted research or development that led to the application for the patent in suit;
(4) where a party has a regular and established physical facility that such party controls and operates, not primarily for the purpose of creating venue, and has—
(A) engaged in management of significant research and development of an invention claimed in a patent in suit prior to the effective filing date of the patent;
(B) manufactured a tangible good that is alleged to embody an invention claimed in a patent in suit; or
(C) implemented a manufacturing process for a tangible good in which the process is alleged to embody an invention claimed in a patent in suit; or
(5) in the case of a foreign defendant that does not meet the requirements of section 1400(b) of title 28, in accordance with section 1391(c)(3) of such title.Further, under proposed Section 330(c) the bill creates a presumption that after a finding of infringement the inventor-owner will suffer irreparable harm unless the court grants an injunction, and provides for rebutting the presumption only upon a showing by clear and convincing evidence that the patentee would not be irreparably harmed.
The bill further provides in Section 330(d) that an inventor-owner can elect, "at any time before final judgment is entered by the court," to obtain recovery under this Section, which will include: (1) profit disgorgement, which will include all infringer's revenues relating to the infringing article, with the infringer bearing the burden of showing any "elements of cost or production" claimed to reduce this amount; (2) interests and costs; (3) treble damages for willful infringement; and (4) attorneys fees in the amount that exceeds 10% of the amounts set forth in subparagraph (1)-(3).
The University of Illinois/Chicago is in Rep. Davis's congressional district but he has not sponsored much patent-related legislation before. One possible consequence of this bill should it pass (which is unlikely) is that patents will remain "owned" by (i.e., assigned) to the named inventors, even in corporate settings, with their corporate masters holding licenses (likely exclusive licenses) to get the benefit of the bill's provisions while retaining traditional control of the patented invention. Care will need to be taken to avoid situations where the scope of the license is so restrictive to amount to an assignment, thus frustrating the purpose (see "St Regis Mohawk Tribe v. Mylan Pharmaceuticals").
According to a press release posted on Rep. Davis's website, "[t]he Inventor Rights Act provides narrowly targeted relief only to inventors that own and control their own patents. This bill does not apply to large corporations, drug companies, or typical non-practicing entities." The bill will be taken up in due course by the House, presumably in the next session beginning in early 2020.
-
By Kevin E. Noonan —
 The Federal Circuit had the opportunity to interpret the extent to which the provisions of 35 U.S.C. § 271(g) require the practice of the patented method that produces a product whose importation imposes infringement liability under the statute to be practiced by a single entity (it does not) in rendering its decision recently in Syngenta Crop Protection, LLC v. Willowood, LLC.
The Federal Circuit had the opportunity to interpret the extent to which the provisions of 35 U.S.C. § 271(g) require the practice of the patented method that produces a product whose importation imposes infringement liability under the statute to be practiced by a single entity (it does not) in rendering its decision recently in Syngenta Crop Protection, LLC v. Willowood, LLC.The case involved accused infringement of U.S. Patent Nos. 5,602,076 (expired February 11, 2014); 5,633,256 (expired February 11, 2014); 5,847,138 (expired December 8, 2015); and 8,124,761 (will not expire until April 15, 2029), directed to fungicides, pesticides, plant growth regulators, comprising pyrimidinyloxy-phenyl acrylates and derivatives, and in particular azoxystrobin. The '076 and '256 patents are directed to compounds, while the '138 and '761 patents are directed to methods for producing these compounds.
Syngenta brought suit for patent infringement and copyright infringement against the Willowood defendants for importation of fungicide formulations comprising azoxystrobin; the copyright infringement assertions were made with regard to Syngenta's label related to "directions for use, storage, and disposal, as well as first-aid instructions and environmental, physical, and chemical hazard warnings." Importantly for the issues in this case, one defendant, Willowood China, produced the accused infringing fungicide in Hong Kong and sold it to Willowood USA, its American affiliate. Willowood USA and another defendant, Willowood LLC then contracted to have third parties formulate the azoxystrobin fungicide and thereafter marketed and sold the product in the U.S. Syngenta alleged infringement of claims 1–4 and 12–14 of the '076 patent, claims 1–3, 5, and 7 of the '256 patent, claims 6 and 12–14 of the '138 patent, and claims 1, 3–5, and 9–10 of the '761 patent. Both parties filed summary judgment motions, upon which the District Court ruled as follows. First, the District Court granted Syngenta's summary judgment motion that Willowood literally infringed claims of the '076 and '256 patents, and that Willowood induced infringement by the LLC affiliate. The District Court denied Syngenta's summary judgment motion that Willowood China literally infringed the '076 and '256 patents, based on the existence of a genuine issues of material fact regarding whether sale occurred in the U.S. or China. The District Court also denied summary judgment of the '138 patent, on the grounds that infringement under 35 U.S.C. § 271(g) required that all steps of a claimed process must be performed by a single infringer (facts regarding this were in dispute). Finally, the District Court denied Syngenta's summary judgment motion regarding infringement of the '761 patent, due to disputed facts on the details of the synthetic methods used by Willowood China. But the District Court granted Syngenta's motion shifting the burden of proof on this issue to Willowood under 35 U.S.C. § 295.
Willowood's summary judgment motion involved Syngenta's copyright claims. The District Court granted this motion based on the provisions of the Federal Insecticide, Fungicide, and Rodenticide Act ("FIFRA"), which precludes label copyright, inter alia, because "much of its labels' text comprises instructions and warnings mandated by FIFRA and EPA regulations, and only limited means of expressing such information exist, extending copyright protection to Syngenta's labels 'would make subsequent labeling practically impossible,'" citing SmithKline Beecham Consumer Healthcare, L.P. v. Watson Pharm. Inc., 211 F.3d 21, 23 (2d Cir. 2000). The United States filed a "statement of interest" supporting Willowood's position.
At trial, the jury entered a verdict that Syngenta did not establish that Willowood China had imported or sold azoxystrobin into the U.S. The jury awarded damages ($76,000) for infringing activity prior to expiration of the '076 and '256 patents by Willowood USA and Willowood LLC. The jury also entered a specific verdict of non-infringement of the '138 patent, based on its determination that Syngenta did not prove that Willowood China performed both steps of the claimed process. Finally, the jury entered a verdict that, under the burden-shifting provisions of the District Court's decision regarding the '761 patent, Willowood had not shown that it did did not synthesize azoxystrobin according to the claimed method and awarded damages ($900,000) for this infringement. This appeal followed, by Willowood on the jury's infringement verdicts and Syngenta on the District Court's grant of summary judgment on its copyright claims.
The Federal Circuit affirmed-in-part, reversed-in-part, vacated-in-part, and remanded, in an opinion by Judge Reyna, joined by Judges Taranto and Stoll. The panel vacated the District Court's grant of summary judgment regarding Syngenta's copyright claims, finding that the District Court had not properly determined whether the provisions of FIFRA conflicted with the Copyright Act, because FIFRA "does not, on its face, require a me-too registrant to copy the label of a registered product" and a conflict will exist only to the extent that Syngenta's label is protected by copyright and is necessary for "expedited approval of Willowood's generic pesticide product." Because the District Court did not reach the merits of Syngenta's copyright claims, the Federal Circuit remanded for the District Court to make this determination. Nevertheless, the Court provided guidance regarding how the purportedly competing goals of FIFRA and the Copyright Act should be resolved:
On remand, the district court should first discern whether the Copyright Act, as interpreted under existing copyright doctrines, would prohibit Willowood's use of any portion of Syngenta's label. The district court should, for instance, consider whether the fair-use doctrine or limits on copyrightable subject matter, such as the merger doctrine, would eliminate infringement. Only if the district court concludes that the Copyright Act would in fact prohibit Willowood's conduct in a manner in- consistent with the purposes of FIFRA should it revisit the question of whether and to what extent FIFRA precludes Syngenta's copyright claims for any part of its pesticide labels.
The opinion then turned to the requirements for showing infringement under § 271(g). In what the opinion characterized as an issue of first impression, the Federal Circuit reversed the District Court's finding that infringement required that "all steps of a patented process be performed by or at the direction or control of a single entity." Agreeing with Syngenta and several amici, the panel held that this requirement was not mandated by the plain meaning of the statutory language. Specifically, the Court stated:
The resolution of this issue turns on the nature of the infringing acts covered by § 271(g). Section 271(g) provides in relevant part that "[w]hoever without authority imports into the United States or offers to sell, sells, or uses within the United States a product which is made by a process patented in the United States shall be liable as an infringer." 35 U.S.C. § 271(g) (2012). This language makes clear that the acts that give rise to liability under § 271(g) are the importation, offer for sale, sale, or use within this country of a product that was made by a process patented in the United States. Id. Nothing in this statutory language suggests that liability arises from practicing the patented process abroad. Rather, the focus is only on acts with respect to products resulting from the patented process. Thus, because the statutory language as a whole is clear that practicing a patented process abroad cannot create liability under § 271(g), whether that process is practiced by a single entity is immaterial to the infringement analysis under that section.
The panel found support for this conclusion by comparison with the language of 35 U.S.C. § 271(a), where the "whoever, without authority" language has been interpreted to impose the single-entity requirement, citing Limelight Networks, Inc. v. Akamai Techs., Inc., 572 U.S. 915, 921–22 (2014). The opinion also relied on the statutory language of § 271(f), where the statutory language includes the requirement that what is induced is combination of components of a claimed article "in a manner that would infringe the patent if such combination occurred within the United States" (emphasis added). The panel stated that "[i]f Congress intended to limit liability under § 271(g) to instances where the patented process was practiced in a manner that would infringe the patent if such practice occurred within the United States—such as it did by requiring a single entity to perform the entire process under § 271(a)—it "kn[ew] precisely how to do so" (which, with regard to § 271(g) Congress clearly did not. And the timing of enactment of the two statutes (§ 271(g) being enacted after § 271(f)) was also significant for the panel's opinion:
"Where Congress includes particular language in one section of a statute but omits it in another section of the same Act, it is generally presumed that Congress acts intentionally and purposely in the disparate inclusion or exclusion," citing Rodriguez v. United States, 480 U.S. 522, 525 (1987) (quoting Russello v. United States, 464 U.S. 16, 23 (1983)).
The panel also relied for its reasoning that the infringing act implicated in § 271(g) was not practice of the claimed method abroad but importation of the product of the patented process. The opinion also found support for its interpretation in the legislative history. Finally, the opinion cited the increased evidentiary burden Willowood's interpretation of the statute would impose on patentees, where manufacture occurs abroad.
Accordingly, the Federal Circuit reversed the District Court's judgment that Willowood USA (whose importation of the accused infringing azoxystrobin was undisputed) did not infringe the '138 patent under § 271(g) and vacated (as not being considered by the District Court) Willowood LLC's infringement liability and remanded. Otherwise, the panel affirmed the District Court in all other respects.
Syngenta Crop Protection, LLC v. Willowood, LLC (Fed. Cir. 2019)
Panel: Circuit Judges Reyna, Taranto, and Stoll
Opinion by Circuit Judge Reyna -
By Kevin E. Noonan —
 The Federal Circuit has grappled with, divisively in some instances, the extent to which the safe harbor provisions of 35 U.S.C. § 271(e)(1) extend to activities not strictly for obtaining regulatory approval, such as post-approval quality testing and "stockpiling" product used for commercial purposes. For example, in Classen Immunotherapies, Inc. v. Biogen IDEC, then-Chief Judge Rader joined by Judge Newman held that "routine" post-approval submissions are outside the safe harbor (over a vigorous dissent by Judge Moore), whereas in the following case, Momenta Pharm. v. Amphastar Pharm., the roles were reversed, with Judge Moore finding herself in the majority (with Judge Dyk), and then-Chief Judge Rader filing a dissent. Yesterday, the Federal Circuit revisited these issues in its decision in Amgen Inc. v. Hospira, Inc.
The Federal Circuit has grappled with, divisively in some instances, the extent to which the safe harbor provisions of 35 U.S.C. § 271(e)(1) extend to activities not strictly for obtaining regulatory approval, such as post-approval quality testing and "stockpiling" product used for commercial purposes. For example, in Classen Immunotherapies, Inc. v. Biogen IDEC, then-Chief Judge Rader joined by Judge Newman held that "routine" post-approval submissions are outside the safe harbor (over a vigorous dissent by Judge Moore), whereas in the following case, Momenta Pharm. v. Amphastar Pharm., the roles were reversed, with Judge Moore finding herself in the majority (with Judge Dyk), and then-Chief Judge Rader filing a dissent. Yesterday, the Federal Circuit revisited these issues in its decision in Amgen Inc. v. Hospira, Inc.The case arose over Amgen's complaint that Hospira infringed its U.S. Patent Nos. 5,865,298 and 5,756,349 relating to cells and methods of preparing erythropoietin (EPO). The jury found that Hospira had not carried its burden of showing either patent to be invalid by clear and convincing evidence; and that Hospira infringed claims 24 and 27 of the '298 patent but had not infringed claims 1-7 of the '349 patent:
'298 patent claims at issue:
24. A method of preparing erythropoietin molecules having a predetermined number of sialic acids per molecule said number selected from the group consisting of 1-14, comprising applying material containing erythropoietin to an ion exchange column and selectively eluting said molecules from the column.
27. A method for obtaining an erythropoietin composition having a predetermined in vivo specific activity comprising preparing a mixture of two or more erythropoietin isoforms of claim 1.
Where independent claim 1 recites:
1. An isolated biologically active erythropoietin isoform having a single isoelectric point and having a specific number of sialic acids per molecule, said number selected from the group consisting of 1-14, and said isoform being the product of the expression of an exogenous DNA sequence in a non-human eucaryotic host cell.
'349 patent claims at issue:
1. Vertebrate cells which can be propagated in vitro and which are capable upon growth in culture of producing erythropoietin in the medium of their growth in excess of 100 U of erythropoietin per 106cells in 48 hours as determined by radioimmunoassay, said cells comprising non-human DNA sequences which control transcription of DNA encoding human erythropoietin.
2. Vertebrate cells according to claim 1 capable of producing in excess of 500 U erythropoietin per 106cells in 48 hours.
3. Vertebrate cells according to claim 1 capable of producing in excess of 1000 U erythropoietin per 106cells in 48 hours.
4. Vertebrate cells which can be propagated in vitro which comprise transcription control DNA sequences, other than human erythropoietin transcription control sequences, for production of human erythropoietin, and which upon growth in culture are capable of producing in the medium of their growth in excess of 100 U of erythropoietin per 106cells in 48 hours as determined by radioimmunoassay.
5. Vertebrate cells according to claim 4 capable of producing in excess of 500 U erythropoietin per 106cells in 48 hours.
6. Vertebrate cells according to claim 4 capable of producing in excess of 1000 U erythropoietin per 106cells in 48 hours.
7. A process for producing erythropoietin comprising the step of culturing, under suitable nutrient conditions, vertebrate cells according to claim 1, 2, 3, 4, 5 or 6.
The jury entered a verdict that Hospira infringed claim 27 of the '298 patent but that Amgen had not shown that Hospira infringed the claims of the '349 patent. The District Court also assessed damages in the amount of $70 million, inclusive of pre- and post-judgment interest.
 Both parties moved for Judgment as a Matter of Law (JMOL). Hospira contended that the jury instructions were "legally erroneous and prejudicial," inter alia, because "ulterior motives and intent are irrelevant to [entitlement to] the Safe Harbor." Specifically, Hospira argued that the instructions did not clarify the use of the terms "use" and "make" as relevant to whether an activity was entitled to the safe harbor provisions of the statute. According to Hospira, the jury was asked to determine whether "manufacture" of the accused infringing article (biosimilar erythropoietin, bEPO) fell under the safe harbor when the only burden Hospira rightfully was obliged to meet was whether "use" of the allegedly infringing bEPO was "reasonably related to obtaining FDA approval." The Court disagreed, stating that "Hospira's potentially infringing 'use' of Amgen' s patented invention is Hospira's manufacture of the EPO drug substance referred to in its BLA (i.e., Hospira's performance of the steps of Amgen's method claims), not Hospira's subsequent use of the EPO drug substance (i.e., Hospira's subsequent use of the product obtained by practicing Amgen's method claims)." Thus, the safe harbor is available to Hospira only if manufacture of its bEPO was "reasonably related to obtaining FDA approval." Hospira's subsequent use of the bEPO is "probative in determining whether Hospira's manufacture of its EPO drug substance was reasonably related to obtaining FDA approval, [but] it is the manufacture itself (not Hospira's subsequent uses of EPO drug substance) that is the potentially infringing act which must be evaluated for safe harbor protection."
Both parties moved for Judgment as a Matter of Law (JMOL). Hospira contended that the jury instructions were "legally erroneous and prejudicial," inter alia, because "ulterior motives and intent are irrelevant to [entitlement to] the Safe Harbor." Specifically, Hospira argued that the instructions did not clarify the use of the terms "use" and "make" as relevant to whether an activity was entitled to the safe harbor provisions of the statute. According to Hospira, the jury was asked to determine whether "manufacture" of the accused infringing article (biosimilar erythropoietin, bEPO) fell under the safe harbor when the only burden Hospira rightfully was obliged to meet was whether "use" of the allegedly infringing bEPO was "reasonably related to obtaining FDA approval." The Court disagreed, stating that "Hospira's potentially infringing 'use' of Amgen' s patented invention is Hospira's manufacture of the EPO drug substance referred to in its BLA (i.e., Hospira's performance of the steps of Amgen's method claims), not Hospira's subsequent use of the EPO drug substance (i.e., Hospira's subsequent use of the product obtained by practicing Amgen's method claims)." Thus, the safe harbor is available to Hospira only if manufacture of its bEPO was "reasonably related to obtaining FDA approval." Hospira's subsequent use of the bEPO is "probative in determining whether Hospira's manufacture of its EPO drug substance was reasonably related to obtaining FDA approval, [but] it is the manufacture itself (not Hospira's subsequent uses of EPO drug substance) that is the potentially infringing act which must be evaluated for safe harbor protection."In addition, Hospira argued that it was unduly prejudicial for the Court not to instruct the jury that "intent is irrelevant to evaluating safe harbor protection." The Court interpreted Hospira's position to be that "intent is entirely irrelevant to the safe harbor analysis" but with that the judge expressly disagreed as being unsupported by the cited case law. Specifically, the Memorandum Opinion interprets Abtox, Inc. v. Exitron Corp., 122 F.3d 1019 (Fed. Cir. 1997), to mean that a party can use the benefit of the safe harbor "to use its data from the tests for more than FDA approval" and "does not look to the underlying purposes or attendant consequences of the activity (e.g., tests led to the sale of the patent), as long as the use is reasonably related to FDA approval" but "did not state that intent was irrelevant in determining whether an activity is reasonably related to obtaining FDA approval." On the contrary, the District Court held that "evidence of intent can be a relevant factor in determining whether an activity is reasonably related to obtaining FDA approval, and that these cases stand for the proposition that evidence of commercial intent is not determinative of the safe harbor inquiry." "[O]nce it is determined that 'the activity is reasonably related to obtaining FDA approval,' . . . intent or alternative uses are irrelevant to its qualification to invoke the section 271(e) shield," according to the District Court. And further:
[A]dopting Hospira's interpretation of the safe harbor defense would expand the defense beyond recognition and create a loophole that would make it virtually impossible to prove infringement in cases involving products regulated by the FDA. Since Hospira's interpretation requires ignoring intent in deciding whether the safe harbor applies, a party could manufacture 200 drug substance batches and earmark them for future use as commercial inventory without infringing, so long as the party used each of those batches for at least one test to generate data of the type used by the FDA in determining whether to approve the drug. In that scenario, each batch would be tested to generate data that could conceivably be used to respond to inquiries from the FDA, making each batch reasonably related to obtaining FDA approval. Essentially, Hospira's interpretation allows a single "token" submission of information derived from a potential infringing act to exempt that act from infringement, without regard to the realities surrounding the potentially infringing act. It seems to me that Hospira's interpretation reads the words "solely" and "reasonably" out of the statute, and that a party's stated intent may be considered as part of whether the manufacture or use of a patented drug was "solely for uses reasonably related to" obtaining FDA approval. I think that the jury instructions properly recited the role of intent in the safe harbor analysis.
The District Court denied both parties' motions for JMOL and this appeal followed.
The Federal Circuit affirmed the jury verdicts and decisions of the District Court with respect to Hospira's appeal and Amgen's cross-appeal, in a decision by Judge Moore joined by Judges Bryson and Chen. The Federal Circuit first held that the District Court correctly construed the claims so that the limitation "a mixture of two or more erythropoietin isoforms of claim 1" did not require mixing of two separately prepared and independently isolated EPO isoforms. This construction was contrary to Hospira's expert testimony, but the District Court and the Federal Circuit disagreed. According to the opinion, "[n]othing in the claim language or the specification suggests that it would be proper to limit claim 27 in the manner Hospira proposes," based in part on disclosure in the '298 patent specification that the claimed mixtures of EPO isoforms could be produced by "isolating selected erythropoietin isoforms simultaneously" (emphasis in opinion). The Federal Circuit concluded that "[t]he specification clearly contemplates the preparation of mixtures of isoforms in more than one way."
The opinion also affirmed based on substantial evidence the jury's finding that Hospira's EPO preparations infringed claim 27 of the '298 patent based on expert testimony and portions of Hospira's aBLA. The Federal Circuit also affirmed the jury's decision that claim 27 was not anticipated by a prior art reference because it did not disclose "a composition with a predetermined in vivo activity" but just that the EPO produced according to the reference was "biologically active."
Regarding the effect of the statutory safe harbor, the opinion rejected Hospira's objections to the jury instructions, particularly the final sentence:
You must evaluate each of the accused activities separately to determine whether the Safe Harbor applies. If you find that an accused activity was reasonably related to the development and submission of information to the FDA for the purpose of obtaining FDA approval, then Hospira has proved its Safe Harbor defense as to that activity. If Hospira has proved that the manufacture of a particular batch was reasonably related to developing and submitting information to the FDA in order to obtain FDA approval, Hospira's additional underlying purposes for the manufacture and use of that batch do not remove that batch from the Safe Harbor defense.
Hospira's objection was that "the jury instructions and verdict form improperly focused the jury on the reasons why each batch of EPO was manufactured, not how each batch was used or whether that use was reasonably related to the development and submission of information to support Hospira's BLA." The opinion rejected this argument, stating "[t]he jury instructions properly articulated the legal principles underlying the Safe Harbor inquiry. Section 271(e)(1)'s exemption from infringement 'extends to all uses of patented inventions that are reasonably related to the development and submission of any information under the FDCA,'" citing Merck KGaA v. Integra Lifesciences I, Ltd., 545 U.S. 193, 202 (2005). The relevant question was whether each of Hospira's acts of manufacturing EPO falling within the scope of claim 27 of the '298 patent "was for uses reasonably related to submitting information to the FDA" and this was the question properly put to the jury by these instructions.
Turning to the merits, the Federal Circuit affirmed the jury's decision that fourteen batches of Hospira's EPO were not protected under the safe harbor. The other seven batches were clearly made for purposes related to obtaining FDA approval (two batches for qualifying Hospira's process and another five for mandatory pre-approval inspections). The opinion found substantial evidence that Hospira planned to use the other batches "[to] serve as commercial inventory to support single dose vial launch stock" and that "[w]hen it resubmitted its application in late 2015 after litigation began, Hospira changed the designation of certain batches from "commercial inventory" to "[continued process verification (CPV)]." Accordingly the Federal Circuit affirmed the jury's determination that these fourteen batches were not protected by the § 271(e)(1) safe harbor.
Nor did the panel find reversible error in the damages awarded to Amgen, which fell between the amounts asserted by Amgen's and Hospira's experts.
Finally, the Federal Circuit affirmed the District Court's denial of Amgen's JMOL on the ground that Amgen had not shown by substantial evidence that Hospira's cells produced EPO in amounts recited in the '349 patent claims.
Amgen Inc. v. Hospira, Inc. (Fed. Cir. 2019)
Panel: Circuit Judges Moore, Bryson, and Chen
Opinion by Circuit Judge Moore -
By Kevin E. Noonan —
 In early November, Senior Party the Broad Institute (along with Harvard University and the Massachusetts Institute of Technology) filed their response to Junior Party the University of California/Berkeley, the University of Vienna, and Emmanuelle Charpentier (collectively, "CVC") Opposition to the Broad's Substantive Motion No. 1 in Interference No. 105,166.
In early November, Senior Party the Broad Institute (along with Harvard University and the Massachusetts Institute of Technology) filed their response to Junior Party the University of California/Berkeley, the University of Vienna, and Emmanuelle Charpentier (collectively, "CVC") Opposition to the Broad's Substantive Motion No. 1 in Interference No. 105,166.Pursuant to the Patent Trial and Appeal Board's rules, the Broad's Reply Brief follows formulaically with Senior Party's point-by-point rebuttal of CVC's grounds for opposition. CVC's first asserted opposition argument was that "judgment estoppel" (quotation marks in opposition brief) does not exist in any form that would prelude this interference. The Broad characterizes this argument as "remarkable," and that it "not only displays a lack of understanding regarding the development of the estoppel rule in interferences, but it also misstates common law estoppel principles." Segueing to the question of "motion estoppel" under Rule 41.127(a), the Broad traces the provisions of this Rule to former Rule 1.658(c), which provided (according to the Broad) for "broad application of estoppel for any type of interference judgment and further listed specific motions that needed to be raised lest motion estoppel apply, including failure to move to add a new claim" (which the Broad argues is the case here). These considerations did not change with revision of the interference rules, according to the Reply Brief, and thus the Board remains empowered under the rules to grant its motion that CVC should be estopped in this interference by the Board's judgment in the prior interference (No. 105,048).
The brief next argues that Rule 127 applies to judgments of no interference-in-fact, contrary to CVC's argument that it cannot because there is no "losing" party under those circumstances, on the basis that there is no express provision in the Rule to this effect. According to the Broad, "the first sentence sets out a general rule, while the second sentence applies it to losing parties and sets out an exception for losing parties"; this does not negate application of estoppel to CVC under the circumstances of this interference, according to the Broad.
The Broad takes direct issue with CVC's argument that the PTAB is powerless to apply estoppel here because doing so would be contrary to "common law estoppel principles," saying bluntly (with reference to Wright's Federal Practice and Procedure treatise) that CVC "misapprehends fundamental principles of claim and issue preclusion," specifically that common law estoppel "foreclos[e] any litigation of matters that never have been litigated, because of a determination that they should have been advanced in an earlier suit." This argument also supports the Broad's rebuttal that 35 U.S.C. § 135(a) "mandates that the PTAB resolve any and all potential priority issues," saying that "the Federal Circuit rejected this argument long ago" citing Eli Lilly & Co. v. Bd. of Regents of Univ. of Washington, 334 F.3d 1264, 1267 (Fed. Cir. 2003). According to the Broad, "[t]he PTAB thus has the discretion and authority to promulgate rules for discontinuing interferences, including estoppel rules that provide consequences for a party's failure to make timely motions."
The brief then reiterates its argument that CVC had the opportunity (to which it did not avail itself) to add a claim to eukaryotic cell embodiments of CRISPR technology in the '048 Interference (pointing out that CVC had indicated in that prior interference that it "might seek authorization to file such a responsive motion"). Having failed to prevail in that interference the Broad characterizes CVC's position in this interference as attempting to get a "second bite at the apple" that the estoppel rules preclude. Further, the Broad disputes CVC's argument that it "could not have moved to add a eukaryotic claim in the '048 Interference because it had no allowed eukaryotic claims at that time," asserting that Standing Order ¶ 208.5.1 permits this. And the Broad rejects CVC's assertion that the Broad could have moved just as well, because Standing Order ¶ 208.5.1 requires the moving party to certify that such a claim was patentable to CVC, which the Broad could not (and certainly would not) do.
Finally in this portion of the brief, the Broad challenges CVC's argument that the "the alleged separate patentability of single-molecule RNA claims" over the Count in the '048 Interference raises a factual issue that precludes the Board from granting the Broad's Substantive Motion No. 1. Here, the Broad argues that this argument is "legally irrelevant," because CVC had the opportunity to file claims to interfering subject matter (i.e., "eukaryotic claims limited to use of single-molecule RNA") but did not.
Returning to the issue of "judgment estoppel," the brief cites MPEP § 2308(b) for the proposition that such estoppel "bars a second interference as to 'claims to the same invention as the count of the interference.'" That is the case here, because "the Broad's single-molecule RNA claims were designated as corresponding to the prior Count," a designation never challenged by CVC.
The brief then sets forth detailed arguments for each of its propositions regarding why CVC's Opposition fails to rebut its arguments in support of Substantive Motion No. 1. To CVC's argument that Rule 127 estoppel does not apply to judgments of no interference-in-fact, the Broad argues that CVC's argument "conflicts with the language of the first sentence and does not give application to both sentences." The first sentence of the Rule, the Broad argues, sets out estoppel without regard to "winners" and "losers," while the second sentence is directed at the "losing" party and setting forth an exception for "any contested subject matter for which that party was awarded a favorable judgment." In the Broad's calculus, neither of these provisions provides CVC with the benefit of pursuing this interference after having failed to pursue subject matter relating to eukaryotic cell embodiments of CRISPR in the '048 Interference. The Broad further cites the Notice of Proposed Rulemaking for Rule 127, 68 Fed. Reg. 66648, 66661 (Nov. 26, 2003), which they argue "flatly contradict[s]" CVC's interpretation of the Rule, and further notes that CVC's argument amounts to an admission that Rule 127 applies to situations where the Board finds there to be no interference-in-fact. Finally on this point the Broad disputes CVC's characterization of the Board's finding of no interference-in-fact as being merely jurisdictional and amounting to dismissal; in their view the Board entered judgment and thus that decisions provokes the estoppel effects set forth in Rule 127.
The brief then sets out an exposition of the Broad's understanding and assertion of the development of the PTAB's estoppel rules, going into detail regarding the similarities and differences between Rule 1.658(c) and Rule 127. In brief, the Broad argues that Rule 1.658(c) applied to "any type of judgment" and that revision of the Rule did not change (and was not intended to change) the scope of this estoppel. And here the brief revisits a familiar refrain, asserting that under Rule 1.658(c) the estoppel would apply for a party that failed to file a motion to add a claim regardless of whether judgment was entered on priority or "ancillary grounds," meaning in this case a finding of no interference-in-fact.
The Broad also argues that these estoppel rules promulgated by the PTAB are not inconsistent with common law principles. Citing Nevada v. United States, 463 U.S. 110, 129-30 (1983), they argue that "common law principles recognize that final judgment in an action results in estoppel with respect to undecided issues that could have been raised but were not" (and further assert that the case CVC cited in support of its position, Duvall v. Atty. Gen. of U.S., 436 F.3d 382, 388 (3d Cir. 2006), is in fact consistent with the Broad's own analysis of estoppel principles in the interference context.
Next, the Broad maintains that there is no statutory requirement that the PTAB always determine priority, based in the Board's discretion whether to declare an interference in the first place (although this argument does not reach the question of whether such a requirement arises once the PTO properly institutes an interference proceeding, although the brief does cite the Director's discretion (but surely not unfettered) to discontinue an interference).
The brief then turns to a disquisition on why CVC's arguments are merely "post-hoc" attempts to be absolved from its failure to move to add eukaryotic CRISPR claims in the '048 Interference. (Indeed, the entirety of the Broad's argument boils down to this point.) The Broad rebuts CVC's contention that its claims were not in condition for allowance as a rationale (amounting to essentially a ripeness argument), based on the absence of any such requirement in Standing Order ¶ 208.5.1 (albeit being an argument of inference rather than directly stated). The Broad further characterizes CVC's arguments as improperly attempting to blame the PTAB for its failure to move, citing colloquy from the teleconference between the parties and the Board regarding authorization to file motions. The Broad states frankly that CVC's argument that the Broad was required to add a claim directed to eukaryotic CRISPR to CVC's application in the '048 Interference was wrong, because Standing Order ¶ 208.5.1 would have required the Broad to "[c]ertify that the movant is not aware of any reason why the claim is not patentable," which they certainly were not willing to do. The brief points out the inconsistency in CVC's arguments regarding prosecution strategy in view of CVC having previously presented eukaryotic CRISPR claims before (and then cancelling them) suggesting the '048 Interference. Finally, the Broad (in a more semantical than legal argument) attempts to rebut CVC's argument that it had not provoked an interference in this case, because by copying the Broad's claims it did so (as opposed to suggesting an interference), which CVC certainly did not do.
The brief ends with two points. First, that this interference is estopped in totality (in opposition to CVC's argument that the scope of any estoppel is limited to dual-molecule CRISPR embodiments), addressing both motion estoppel and judgment estoppel, and restating succinctly its argument in favor of estoppel:
The PTAB's rules afforded CVC every opportunity to present claims and arguments regarding the eukaryotic subject matter of the 048 Interference. The PTAB's rules do not allow a second run at another party's claims to that same subject matter in a later interference. Indeed, allowing otherwise would vitiate the goals of finality and efficiency served by the estoppel rules.
And of course the Broad does not miss its opportunity to state unequivocally that "CVC's claims should not issue" in the event that this interference should be dissolved based on the Broad's own arguments about estoppel:
CVC's argument ignores that Broad has already been awarded claims to the eukaryotic invention in issued patents. CVC cannot obtain claims to the same subject matter absent an interference with Broad. CVC could have had such an interference previously but strategically declined to give the PTAB the option of implementing such an interference at that time. Accordingly, CVC is estopped from pursuing such an interference, and cannot obtain Broad's eukaryotic claims.
-
By Donald Zuhn —
 Earlier this fall, Athena Diagnostics, Inc.; Oxford University Innovation Ltd.; and Max-Planck-Gesellschaft Zur Forderung Der Wissenschaften E.V. ("Athena") filed a petition for a writ of certiorari in Athena Diagnostics, Inc. v. Mayo Collaborative Services, LLC, requesting that the Supreme Court "provide much-needed guidance on the important question of the patent eligibility of medical diagnostic tests." Athena begins its petition by stating that:
Earlier this fall, Athena Diagnostics, Inc.; Oxford University Innovation Ltd.; and Max-Planck-Gesellschaft Zur Forderung Der Wissenschaften E.V. ("Athena") filed a petition for a writ of certiorari in Athena Diagnostics, Inc. v. Mayo Collaborative Services, LLC, requesting that the Supreme Court "provide much-needed guidance on the important question of the patent eligibility of medical diagnostic tests." Athena begins its petition by stating that:Across eight opinions concurring or dissenting in the denial of rehearing en banc, the Federal Circuit unanimously agreed that the claims to a medical diagnostic method in this case should be patent-eligible. But the court split 7-5 on whether this Court's precedent foreclosed such a result or whether it was the Federal Circuit's own misinterpretation of that precedent that has denied patent protection to diagnostic tests. Numerous judges asked this Court to provide guidance.
Athena then sets forth the question presented as:
Whether a new and specific method of diagnosing a medical condition is patent-eligible subject matter, where the method detects a molecule never previously linked to the condition using novel man-made molecules and a series of specific chemical steps never previously performed.
At issue in the case are claims 6-9 of U.S. Patent No. 7,267,820, which is directed to methods for diagnosing neurological disorders by detecting antibodies to a protein called muscle-specific tyrosine kinase (MuSK). The '820 patent discloses that about 80% of patients with Myasthenia gravis (MG) produce acetylcholine receptor autoantibodies, and that the remaining 20% do not. The named inventors of the '820 patent discovered that many of the 20% of MG patients without acetylcholine receptor autoantibodies instead generate autoantibodies to MuSK. The '820 patent contains only one independent claim, which, while not at issue in the case, recites:
1. A method for diagnosing neurotransmission or developmental disorders related to muscle specific tyrosine kinase (MuSK) in a mammal comprising the step of detecting in a bodily fluid of said mammal autoantibodies to an epitope of muscle specific tyrosine kinase (MuSK).
Claims 6-9, which are at issue in the case, recite:
6. A method according to claim 3 whereby the intensity of the signal from the anti-human IgG antibody is indicative of the relative amount of the anti-MuSK autoantibody in the bodily fluid when compared to a positive and negative control reading.
7. A method according to claim 1, comprising contacting MuSK or an epitope or antigenic determinant thereof having a suitable label thereon, with said bodily fluid, immunoprecipitating any antibody/MuSK complex or antibody/MuSK epitope or antigenic determinant complex from said bodily fluid and monitoring for said label on any of said antibody/MuSK complex or antibody/MuSK epitope or antigen determinant complex, wherein the presence of said label is indicative of said mammal is suffering from said neurotransmission or developmental disorder related to muscle specific tyrosine kinase (MuSK).
8. A method according to claim 7 wherein said label is a radioactive label.
9. A method according to claim 8 wherein said label is 125I.
Athena markets a test called FMUSK that functions by evaluating those antibodies. After Mayo developed two competing tests, Athena filed suit against Mayo for infringement of the '820 patent, and Mayo moved to dismiss under Rule 12(b)(6), arguing that the asserted claims of the '820 patent were invalid under § 101 for claiming patent ineligible subject matter. The District Court granted Mayo's motion, and Athena appealed for a determination of whether claims 6-9 are patent eligible under § 101.
In February, a divided Federal Circuit panel affirmed a decision by the District Court, holding claims 6-9 invalid under 35 U.S.C. § 101, and dismissing Athena's complaint for infringement under Fed. R. Civ. P. 12(b)(6). In the panel opinion, authored by Judge Lourie and joined by Judge Stoll (with Judge Newman dissenting), the panel majority "ultimately agree[d] with Mayo that, under Mayo [Collaborative Services v. Prometheus Laboratories, Inc., 566 U.S. 66, 70 (2012)], the claims are directed to a natural law," and that in the instant case, the natural law "is the correlation between the presence of naturally-occurring MuSK autoantibodies in bodily fluid and MuSK related neurological diseases like MG." However, the majority also explained in a footnote that:
[I]n our view, providing patent protection to novel and non-obvious diagnostic methods would promote the progress of science and useful arts. But, whether or not we as individual judges might agree or not that these claims only recite a natural law, . . . the Supreme Court has effectively told us in Mayo that correlations between the presence of a biological material and a disease are laws of nature, and "[p]urely 'conventional or obvious' '[pre]-solution activity' is normally not sufficient to transform an unpatentable law of nature into a patent-eligible application of such a law," . . . . Our precedent leaves no room for a different outcome here [citations omitted].
In July, the Federal Circuit issued a per curiam Order in the case, denying Athena's petition for rehearing en banc. The per curiam Order was accompanied by eight opinions, four concurring in the denial of the petition and four dissenting in the denial of the petition. The four concurring opinions were authored by Circuit Judges Lourie, Hughes, Dyk, and Chen, with Chief Judge Prost and Circuit Judges Reyna, Taranto, and Hughes joining in one of the concurrences and Circuit Judge Chen joining in one concurrence and several parts of another. The four dissenting opinions were authored by Circuit Judges Moore, Newman, Stoll, and O'Malley, with Circuit Judges O'Malley and Stoll joining in one of the dissents and Circuit Judge Wallach joining in three of the dissents.
 In its petition for a writ of certiorari, Athena declares that "[a]cross eight separate opinions in which the court divided 7-5 on denying en banc review, the Federal Circuit issued an unprecedented cry for help from this Court to clarify the patent eligibility of medical diagnostic tests." Athena then requests that the Supreme Court "heed that cry and provide much-needed guidance on the proper application of the judicially-created exceptions to Section 101 of the Patent Act." Noting that since the Supreme Court's decision in Mayo, "the Federal Circuit has invalidated every diagnostic claim to come before it as ineligible subject matter for patent protection," Athena points out that the Federal Circuit, while denying Athena's petition for rehearing en banc, "agreed with Athena that sufficiently specific diagnostic methods with proven utility like the ones here should be patent-eligible," and that there was broad consensus among the Federal Circuit judges that Supreme Court review was "desperately needed."
In its petition for a writ of certiorari, Athena declares that "[a]cross eight separate opinions in which the court divided 7-5 on denying en banc review, the Federal Circuit issued an unprecedented cry for help from this Court to clarify the patent eligibility of medical diagnostic tests." Athena then requests that the Supreme Court "heed that cry and provide much-needed guidance on the proper application of the judicially-created exceptions to Section 101 of the Patent Act." Noting that since the Supreme Court's decision in Mayo, "the Federal Circuit has invalidated every diagnostic claim to come before it as ineligible subject matter for patent protection," Athena points out that the Federal Circuit, while denying Athena's petition for rehearing en banc, "agreed with Athena that sufficiently specific diagnostic methods with proven utility like the ones here should be patent-eligible," and that there was broad consensus among the Federal Circuit judges that Supreme Court review was "desperately needed."Athena provides three reasons for the Supreme Court to grant its petition. First, Athena suggests that the Court should review the case to clarify the Court's § 101 precedent. Second, Athena contends that the Federal Circuit's holding threatens to disrupt important medical innovation. And finally, Athena asserts that the case provides an ideal vehicle to clarify the patent eligibility of diagnostic methods.
With respect to the first reason, Athena argues that:
The Federal Circuit is deeply divided on how to apply this Court's precedent. Every judge of the en banc court was troubled by the conclusion that Athena's claims are not patent-eligible, despite their use of specific chemical steps and man-made molecules in the first diagnostic test ever developed for a large number of patients with MG. The disagreement focused on whether this Court's decision in Mayo Collaborative Services v. Prometheus Laboratories, Inc., 566 U.S. 66 (2012) mandates such an unfortunate result, or whether, as five judges argued, the Federal Circuit itself created the problem by misinterpreting this Court's precedent. Either way, the fractured decisions of the Federal Circuit are a clarion call for this Court to grant certiorari and provide much-needed guidance on the important question of the patent eligibility of medical diagnostic tests.
After pointing to other decisions in which "the Federal Circuit expressed confusion and division on patent eligibility or called for this Court's intervention," Athena states that:
The collective and consistent cry for help from the Federal Circuit, culminating in this case, is extraordinary and emphasizes just how critical this Court's guidance is. Patent-eligibility is a threshold question of enormous importance to innovation and the economy, and yet the Federal Circuit has badly misinterpreted the judicially-created exceptions to the statute to bar patent protection to inventions that meet all the statutory criteria for patenting, including the requirements that claims be novel and nonobvious. The judges tasked with hearing all patent appeals in the United States have now told this Court in no uncertain terms that they are confused and need clarification on how to apply those judicial exceptions. The Court should heed that call.
Athena provides five points of confusion that it contends have emerged "as courts and litigants have struggled to apply—or have misapplied—the judicially-created exceptions to Section 101." First, Athena contends that "courts have struggled with the tension between the Federal Circuit's striking down a diagnostic method claim that uses novel man-made molecules and this Court's holding that a 'molecule that is not naturally occurring' is 'not a 'product of nature' and is patent eligible under § 101.'" Second, Athena contends that "courts have struggled to apply the principles articulated in the context of the unusual facts of Mayo to more typical patent claims," arguing that in contrast with the claims at issue in the instant case, "[t]he claim in Mayo was an oddity" because the two actions required by the claim were steps that were already performed by doctors before the patent was issued. Third, Athena argues that "courts have struggled with the level of abstraction at which to determine whether the steps of a claim 'transform an unpatentable law of nature into a patent eligible application of such a law.'" Fourth, Athena asserts that "the Federal Circuit's approach gives little consideration to the 'preemptive' scope of a claim, contrary to this Court’s guidance," stating that the Federal Circuit "has transformed the preemption analysis into a one-way ratchet" in which claims that do not prevent others from using a natural law are nevertheless deemed to be patent ineligible. Finally, Athena argues that "courts have struggled with what it means to review a claim 'as a whole.'"
Athena suggests that "[g]iven the lower courts' widespread confusion about the judicially-created exceptions to patent eligibility, government officials, practitioners, and commentators have all echoed and amplified the need for guidance from this Court." Athena argues that "[t]he Federal Circuit's wavering hand has only made the PTO's struggle implementing this Court's Section 101 case law more difficult." Athena therefore contends that the Supreme Court "should grant review to provide the guidance that the Federal Circuit, the PTO, practitioners, and scholars all agree is urgently needed."
Moving on to the second reason for the Supreme Court to grant its petition, Athena contends (citing Judges Moore and Lourie) that the Federal Circuit's decision in this case has effectively shut the door for good for the vast majority of diagnostic claims, "establishing a 'per se rule' that bars 'diagnostic kits and techniques,' . . . insofar as they are deemed to 'consist of routine steps to observe the operation of a natural law,' . . . no matter how novel or useful they are." Athena declares that "[t]his rule will have devastating consequences," noting that in the month after Mayo was decided, the rejection rate for medical diagnostics applications rose from 7% to 32%, and that by the time that Alice Corp. v. CLS Bank Int'l was decided, the rejection rate increased to 50%, eventually rising to 64%. According to Athena, "[w]ith the decision below, that rate will climb significantly higher—if inventors even bother to apply for patents for diagnostic methods at all." Athena argues that such an outcome will be devastating for U.S. healthcare because diagnostics, which account for only 2.3% of national healthcare spending, reduce healthcare costs by allowing doctors to replace expensive late-stage therapies with cost-effective early-stage therapies.
Athena concludes its petition by arguing that this case provides an ideal vehicle to clarify the patent eligibility of diagnostic methods. In particular, Athena argues that its claims "recite a new and specific method of diagnosing MG through a series of concrete laboratory steps, so that patients can be accurately and expeditiously diagnosed," declaring that "[i]f these claims are patent-ineligible, that is truly the end for most medical diagnostic methods." Athena then points to several ways in which this case would provide an opportunity to clarify the patent eligibility of medical diagnostic tests.
First, Athena argues that its claims provide an opportunity to clarify the law with respect to the use of novel man-made molecules. According to Athena, the use of man-made compounds in its claimed methods "makes this an easy case," since "[t]here is no reason in law or logic that a claim that goes even further [than reciting man-made molecules] and limits itself to specific uses of novel man-made compounds should lose its eligibility for patent protection" (emphasis in opinion).
Athena next suggests that its claims provide an opportunity to clarify the law with respect to the recitation of specific chemical steps to achieve a new and useful result. Arguing that "[t]he patent claims here are a world apart from the claim invalided in Mayo," Athena notes that while the inventors made an undisputedly groundbreaking discovery that 20% of MG patients develop the disorder because they generate autoantibodies to MuSK, "that is not what they claimed." Instead, Athena argues that the inventors claimed an application of that discovery.
Athena closes its petition by arguing that "the rules the Federal Circuit has relied on to invalidate medical diagnostic claims find no support in the statute," adding that "[t]he language chosen by Congress easily encompasses Athena's claims," and that "[t]his case thus turns entirely on judicially-created exceptions, unsupported by the statutory text." Athena states that:
This Court, as the creator of those non-textual exceptions, bears a special responsibility to ensure that they are properly interpreted and applied. The Court has admonished courts to "tread carefully" lest the exceptions "swallow all of patent law." Alice, 573 U.S. at 217. But the Federal Circuit has not heeded that admonition, allowing those exceptions to expand ever outward and swallow the field of medical diagnostics. The legal issues are too fundamental, and the stakes too high, to allow that misapplication of the law to stand. The Court should grant the petition and reverse.
For additional information regarding this topic, please see:
• "The Proper Role of the Federal Circuit," July 25, 2019
• "Athena Diagnostics v. Mayo Collaborative Services — The Dissents," July 17, 2019
• "Athena Diagnostics v. Mayo Collaborative Services — The Concurrences," July 14, 2019
• "Athena Diagnostics, Inc. v. Mayo Collaborative Services, LLC (Fed. Cir. 2019)," July 9, 2019
• "Invitation to Join Amicus Brief in Athena Diagnostics v. Mayo," April 11, 2019
• "Athena Diagnostics, Inc. v. Mayo Collaborative Services, LLC (Fed. Cir. 2019)," February 12, 2019
• "Athena Diagnostics, Inc. v. Mayo Collaborative Services, LLC (D. Mass. 2017)," August 8, 2017 -
By Kevin E. Noonan —
 As previously discussed, Senior Party The Broad Institute (joined by Harvard University and MIT) on October 14th filed Substantive Motion No 2 (to substitute the count) in the current interference over CRISPR technology (No. 106,115).
As previously discussed, Senior Party The Broad Institute (joined by Harvard University and MIT) on October 14th filed Substantive Motion No 2 (to substitute the count) in the current interference over CRISPR technology (No. 106,115).Count 1 of the interference as declared is:
An engineered, programmable, non-naturally occurring Type II CRISPR-Cas system comprising a Cas9 protein and at least one guide RNA that targets and hybridizes to a target sequence of a DNA molecule in a eukaryotic cell, wherein the DNA molecule encodes and the eukaryotic cell expresses at least one gene product and the Cas9 protein cleaves the DNA molecules, whereby expression of the at least one gene product is altered; and, wherein the Cas9 protein and the guide RNA do not naturally occur together, wherein the guide RNAs comprise a guide sequence fused to a tracr sequence.
or
A eukaryotic cell comprising a target DNA molecule and an engineered and/or non-naturally occurring Type II Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR)-— CRISPR associated (Cas) (CRISPR-Cas) system comprising
a) a Cas9 protein, or a nucleic acid comprising a nucleotide sequence encoding said Cas9 protein; and
b) a single molecule DNA-targeting RNA, or a nucleic acid comprising a nucleotide sequence encoding said single molecule DNA-targeting RNA; wherein the single molecule DNA-targeting RNA comprises:
i) a targeter-RNA that is capable of hybridizing with a target sequence in the target DNA molecule, and
ii) an activator-RNA that is capable of hybridizing with the targeter-RNA to form a double-stranded RNA duplex of a protein- binding segment,
wherein the activator-RNA and the targeter-RNA are covalently linked to one another with intervening nucleotides; and
wherein the single molecule DNA-targeting RNA is capable of forming a complex with the Cas9 protein, thereby targeting the Cas9 protein to the target DNA molecule, whereby said system is capable of cleaving or editing the target DNA molecule or modulating transcription of at least one gene encoded by the target DNA molecule.The Broad's proposed Count 2 is:
A method, in a eukaryotic cell, of cleaving or editing a target DNA molecule or modulating transcription of at least one gene encoded by the target DNA molecule, the method comprising:
contacting, in a eukaryotic cell, a target DNA molecule having a target sequence with an engineered and/or non-naturally-occurring Type II Clustered Regularly lnterspaced Short Palindromic Repeats (CRISPR)-CRISPR associated Cas) (CRISPR-Cas) system comprising:
a) a Cas9 protein, and
b) RNA comprising
i) a targeter-RNA that is capable of hybridizing with the target sequence of the DNA molecule or a first RNA comprising (A) a first sequence capable of hybridizing with the target sequence of the DNA molecule and (B) a second sequence; and
ii) an activator-RNA that is capable of hybridizing to the targeter-RNA to form an RNA duplex in the eukaryotic cell or a second RNA comprising a tracr sequence that is capable of hybridizing to the second sequence to form an RNA duplex in the eukaryotic cell,
wherein, in the eukaryotic cell, the targeter-RNA or the first sequence directs the Cas9 protein to the target sequence and the DNA molecule is cleaved or edited or at least one product of the DNA molecule is altered.The distinction the Broad made in its Motion is between embodiments of CRISPR methods that are limited to "single-molecule guide RNA" (aka "fused" or "covalently linked" species), versus embodiments that encompass single-molecule and "dual molecule" species (wherein the in the latter versions the "targeter-RNA" and "activator-RNA" as recited in the proposed Count are not covalently linked). The Broad argued that this Count should be adopted by the Board because it "properly describes the full scope of the interfering subject matter between the parties because both parties have involved claims that are generic, non-limited RNA claims." The brief also argued that proposed Count 2 "sets the correct scope of admissible proofs [i.e., their own] for the breakthrough invention described by the generic claims at issue in these proceedings—the successful adaption of CRISPR-Cas9 systems for use in eukaryotic environments," which The Broad contended current Court 1 (in either alternative) does not.
 Early last month (November 7th), Junior Party the University of California, Berkeley; the University of Vienna; and Emmanuelle Charpentier; collectively, "CVC") filed its Responsive Motion (Contingent) to the Broad's motion no. 2, to be awarded priority to its earlier applications for the subject matter of proposed new Count 2. (Careful readers with appreciate that in many ways this brief is substantially identical to CVC's Substantive Motion No. 1 filed to be accorded benefit to the same priority applications for the current Count.) As required by the rather formulaic procedural rules of interferences, the brief is structured to address (and rebut) Senior Party's argument; the argument (simply stated) is that CVC is entitled to priority to the following priority applications: USSN 61/652,086, filed May 25, 2012 (P1); USSN 61/716,256, filed October 19, 2012 (P2): USSN 61/757,640, filed January 28, 2013 (P3); USSN 13/842,859, filed March 15, 2013; USSN 14/685,504, filed April 13, 2015; or USSN 15/138,604, filed April 26, 2016.
Early last month (November 7th), Junior Party the University of California, Berkeley; the University of Vienna; and Emmanuelle Charpentier; collectively, "CVC") filed its Responsive Motion (Contingent) to the Broad's motion no. 2, to be awarded priority to its earlier applications for the subject matter of proposed new Count 2. (Careful readers with appreciate that in many ways this brief is substantially identical to CVC's Substantive Motion No. 1 filed to be accorded benefit to the same priority applications for the current Count.) As required by the rather formulaic procedural rules of interferences, the brief is structured to address (and rebut) Senior Party's argument; the argument (simply stated) is that CVC is entitled to priority to the following priority applications: USSN 61/652,086, filed May 25, 2012 (P1); USSN 61/716,256, filed October 19, 2012 (P2): USSN 61/757,640, filed January 28, 2013 (P3); USSN 13/842,859, filed March 15, 2013; USSN 14/685,504, filed April 13, 2015; or USSN 15/138,604, filed April 26, 2016.Mirroring the Broad's motion, CVC sets forth in detail the disclosure in its earlier priority applications for at least one embodiment falling within the scope of proposed Count 2. And as the Broad did in its motion, the brief hagiograhically recites the "ground-breaking" nature of their work, stating that the earliest priority document (P1) disclosed "the minimal components required to generate a functional CRISPR-Cas9 DNA-cleavage complex—Cas9, crRNA, and tracrRNA." In addition, and addressing the Broad's argument that CVC's disclosure (and this interference) were directed to single-molecule embodiments of CRISPR, CVC argues that on the contrary this priority document "disclosed, for the first time, that complexes of Cas9 and a double- or single-molecule DNA-targeting RNA . . . are useful for targeted DNA cleavage and described numerous applications of this gene-editing technology, including modifying target DNA in eukaryotic cells" and that "[t]he CVC inventors immediately understood that the CRISPR-Cas9 DNA-cleavage complex could be used in a variety of different cellular and noncellular settings." The brief recites (prophetic) Example 1 in the P1 specification, asserting that the failure of the P1 specification to show actual reduction to practice is not required to satisfy the requirement for entitlement benefit. CVC also cautions the Board against any attempt by the Broad to "erroneously to link the issues in this motion to the PTAB's termination of Interference No. 106,048 due to no interference-in-fact," stating that "the legal and factual issues raised here are fundamentally different from those decided in the prior '048 proceeding" based on the PTAB's own prior statements of the grounds for its no interference-in-fact determination. Rather, according to CVC:
[A person of ordinary skill in the art] reading P1 in light of the state of the art at the time of filing would have understood that the application describes and enables at least one embodiment within the scope of Proposed Count 2. Moreover, post-filing-date publications report successfully practicing CVC's claimed invention in eukaryotes using the very methods and components that P1 describes. The Board should therefore accord CVC the benefit of P1's May 25, 2012 filing date with respect to Proposed Count 2.
What follows is a succinct statement of the Precise Relief Requests (pursuant to PTAB rules) and support in the P1 specification for this relief, recited in the alternative with the other priority documents recited in CVC's request for relief. The standard, undisputed by the parties, is that to be accorded benefit of priority a prior application must show constructive reduction to practice (CRTP) regarding at least one embodiment falling within the scope of the count, citing Falkner v. Inglis, 448 F.3d 1357, 1362 (Fed. Cir. 2006). After setting out the legal grounds for CRTP, the brief then applies these rubrics to the disclosure in P1 for subject matter falling within the scope of Count 2 (which CVC argues satisfies these requirements). Along the way, the brief also suggests that "Broad will doubtlessly rely on cherry-picked quotes about whether or not the inventors or experts knew CRISPR would work in eukaryotic cells before testing it," rejecting these anticipated arguments on the ground that CTRP is grounded on what is disclosed in the specification. The relevant P1 disclosure for CVC is that CRISPR is functional "when removed from its natural prokaryotic cellular milieu, which is highly relevant here because it establishes CVC's possession of the necessary and sufficient components for a functional CRISPR-Cas9 DNA-cleavage complex regardless of its environment" (emphasis in brief).
The argument is illustrated by Figures from the P1 application:

And explicit disclosure compared with the elements of proposed Count 2.The brief then sets forth specific disclosure related to the elements of Count 2 (also provided in Appendix 3 entitled "Exemplary Evidence of Constructive Reduction to Practice of Proposed Count 2 in the P1 '068 Application"), and CVC further asserts that "[t]he inventors fully grasped the broad utility of such a method as aptly illustrated by the many types of 'target cells of interest' suitable for the methods described in P1, including a variety of eukaryotic cells (elements [1]-[2]) such as a fish, human, and fruit fly cell," noting that "[t]hese features are not merely recited in P1, but diagrammed, discussed, and specifically exemplified showing the inventors' possession" based on express disclosure cited with particularity in the brief (including specifically the prophetic use of CRISPR in fish cells). CVC presents explicit argument relating to what the skilled worker would understand CVC possessed and would be able to accomplish without undue experimentation with regard to the CRTP requirement for being accorded benefit. Finally in this regard, the brief asserts that post-filing evidence (much of it by third parties) further supports a conclusion of constructive reduction to practice in the P1 disclosure:
Within a year of CVC publishing that Cas9 and a [single guide RNA] such as chimera A (or a double-molecule RNA) formed a functional DNA-cleavage complex (Ex. 3202), other researchers used materially the same components and methods disclosed in P1 to practice the fish cell embodiment—objectively confirming that P1 enables practicing the method of E4. While the studies described below published after P1's May 25, 2012 filing
 date, post-priority date evidence "may show, for example, that practicing the invention did not require undue experimentation," citing Amgen, Inc. v. Sanofi, 872 F.3d 1367, 1379 (Fed. Cir. 2017); see also, In re Wands, 858 F.2d 731, 739 (Fed. Cir. 1988); In re Hogan, 559 F.2d 595, 605 (Fed. Cir. 1977).
date, post-priority date evidence "may show, for example, that practicing the invention did not require undue experimentation," citing Amgen, Inc. v. Sanofi, 872 F.3d 1367, 1379 (Fed. Cir. 2017); see also, In re Wands, 858 F.2d 731, 739 (Fed. Cir. 1988); In re Hogan, 559 F.2d 595, 605 (Fed. Cir. 1977).CVC supports its allegations of what the skilled person would understand from the disclosure in the P1 specification by declaration testimony an expert, Dr. Peterson (who eventually will be subject to cross-examination by the Broad in this interference). Specifically, Dr. Peterson attests extensively to the applicability of CRISPR as set forth in P1 to use in human cells, which CVC asserts is also shown by third-party success in applying CRISPR to human cells, and fruit fly cells. Thus, CVC asserts "[i]n sum, the great weight of evidence compels a finding that P1 describes and enables each of the fish (E4), human (E5), and fruit fly (E6) cell methods, any one of which, on its own provides a CRTP of Proposed Count 2."
The brief then turns on the distinction between what was required for the Broad to prevail (as it did) in the earlier interference and in this one:
Broad's previously asserted arguments wrongly impose a reasonable expectation of success standard (obviousness) on §112, first paragraph, which contains no such standard. Obviousness "turns on . . . whether the claimed invention would have been obvious in view of the prior art." Allergan, Inc. v. Sandoz, Inc., 796 F.3d 1293, 1310 (Fed. Cir. 2015) (emphasis in original). "In contrast, the enablement inquiry turns on whether the skilled artisan, after reading the specification, would be able to make and use the claimed invention without undue experimentation." Id. (emphasis in original). Similarly, "[t]he standard for satisfying the written description requirement is whether the disclosure 'allow(s) one skilled in the art to visualize or recognize the identity of the subject matter purportedly described.'" Alcon, 745 F.3d at 1190 (emphasis added; citation omitted); see also, Ariad, 598 F.3d at 1351. Any argument to the contrary erroneously conflates obviousness with written description and enablement.
Finally, the brief argues in the alternative that the other applications CVC asserts for priority contain the P1 disclosure relied upon for priority in this brief, and thus for the same reasons (set forth in brief for each reference) CVC is entitled to priority to these applications, based on the "continuous chain" of priority in these CVC applications.
-
Supreme Court Rejects USPTO's Attempt to Recover Attorney's Fees in All District Court "Appeals" from PTAB Decisions
By Joshua Rich —
 In Peter v. NantKwest, Inc., decided by the U.S. Supreme Court earlier today, the Court considered whether the U.S. Patent and Trademark Office could compel an aggrieved applicant to pay its attorney's fees in a § 145 review, regardless of who wins or loses. The Court unanimously rejected the USPTO's arguments. In doing so, the Court bolstered the "American Rule" on attorney's fees and preserved § 145 proceedings as a financially viable option for review of adverse Patent Trial and Appeal Board ("PTAB") decisions. The decision expressly considered only patent reviews, but it also applies to analogous reviews of trademark decisions. Thus, it has impact on cases far more broadly than just § 145 proceedings.
In Peter v. NantKwest, Inc., decided by the U.S. Supreme Court earlier today, the Court considered whether the U.S. Patent and Trademark Office could compel an aggrieved applicant to pay its attorney's fees in a § 145 review, regardless of who wins or loses. The Court unanimously rejected the USPTO's arguments. In doing so, the Court bolstered the "American Rule" on attorney's fees and preserved § 145 proceedings as a financially viable option for review of adverse Patent Trial and Appeal Board ("PTAB") decisions. The decision expressly considered only patent reviews, but it also applies to analogous reviews of trademark decisions. Thus, it has impact on cases far more broadly than just § 145 proceedings.A patent applicant dissatisfied with a PTAB decision[1] has two options for review of that decision. First, the applicant can appeal the decision to the U.S. Court of Appeals for the Federal Circuit under 35 U.S.C. § 141. In such cases — which are the vast majority of reviews of PTAB decisions — the Federal Circuit considers only the record before the PTAB and reviews that record deferentially for administrative correctness. Second, the applicant can also file a civil action in the U.S. District Court for the Eastern District of Virginia under 35 U.S.C. § 145 and introduce new evidence for consideration de novo. One downside of the second route, however, is that "[a]ll of the expenses of the proceeding shall be paid by the applicant." In NantKwest, Inc. v. Lee, which later became Peter v. NantKwest, the USPTO argued for the first time that those expenses included the salaries of the USPTO attorneys and paralegals trying the case, regardless of the outcome of the case.
The Supreme Court's starting point in considering the USPTO's argument was the "American Rule" on attorney's fees: "Each litigant pays his own attorney's fees, win or lose, unless a statute or contract provides otherwise." The USPTO argued that the American Rule didn't apply to § 145 proceedings because fee-shifting statutes usually reward only prevailing parties, and § 145 doesn't require the USPTO to be a prevailing party in order to recover expenses. But the Court noted that there are exceptions to the prevailing party requirement, such as cases brought under the National Childhood Vaccine Injury Act; those exceptions are—and are required to be—extremely clear in order to create an exception to the American Rule.
Here, the Court found § 145 provides no such clarity. The word "expenses" itself is ambiguous as to whether it would include attorney's fees. Next, the phrase "expenses of the proceeding" is similar to the term of art "expensæ litis" (Latin for expenses of the litigation), which traditionally has excluded attorney's fees. And the word "all" preceding "expenses of the proceeding" does not broaden the type of expenses recoverable, just the extent to which appropriate expenses are recoverable. Thus, the American Rule applies, and the USPTO cannot recover its attorney's fees as expenses as a matter of right under § 145.
To the extent that there was any question remaining as to whether the USPTO's attorney's fees were expenses under § 145, the Court considered both statutory usage of the term "expenses" in other contexts and the USPTO's history of conduct in § 145 actions. The terms "expenses" and "attorney's fees" appear in tandem numerous times in statutes, suggesting that the former does not always include the latter. When expenses are defined as including attorney's fees, however, the definition is clear (in a way that it is not under § 145). Perhaps most damning to the USPTO's case, however, was its own course of conduct. For over 170 years, until the NantKwest case, the USPTO never argued that expenses under § 145 included attorney's fees. Thus, neither the Patent Act nor other statutes provided a basis to vary from the American Rule. The USPTO cannot recover its attorney's fees as a matter of right as expenses in a § 145 proceeding.
Although § 145 proceedings are quite rare, the Court's decision is important in preserving them as an option under any circumstances. Applicants often appeal under § 141 not only because the record may be complete, but also because it is quicker and less costly. The cost distinction would be even more extreme if the applicant had to pay the USPTO's attorney's fees, which could have easily doubled the cost of "expenses" due under the law. The financial difference would have, as a practical matter, made § 145 review financially unfeasible.
For the Court, preserving the American Rule was also a key concern. There are circumstances in which the American Rule does not apply in patent cases but those situations almost always arise under 35 U.S.C. § 285, which clearly allows for an award of attorney's fees.[2] To allow attorney's fees to be collected under § 145, where the entitlement to fees is far less clear, would lower the bar for the recovery of fees under other statutes as well. The ultimate effect would be a watering down of the American Rule, and a resulting increase in collateral litigation over attorney's fees.
Finally, the Trademark Act has procedures for review parallel to §§ 141 and 145. In fact, a trademark case decided under the statute parallel to § 145, Shammas v. Focarino, 784 F.3d 219 (4th Cir. 2015), served as the strongest, most analogous precedent for the Federal Circuit's panel decision below awarding the USPTO attorney's fees automatically. Today's Supreme Court decision undermines the reasoning of the Shammas case, and therefore should apply with full force to district court review of Trademark Trial and Appeal Board decisions as well.
Peter v. NantKwest, Inc. (2019)
Opinion by Justice Sotomayor, joined by Chief Justice Roberts and Justices Thomas, Ginsburg, Breyer, Alito, Kagan, Gorsuch, and Kavanaugh[1] The procedures identified here, and the holding in the NantKwest case, apply only to the review of prosecution decisions, not post-grant proceedings such as inter partes review.
[2] Section 285 provides: "The court in exceptional cases may award reasonable attorney fees to the prevailing party." Attorney’s fees are also sometimes awarded under Rule 11 of the Federal Rules of Civil Procedure, which has a similarly clear provision on the award of fees, or the Court’s inherent sanctions power.
-
By Kevin E. Noonan —
 On October 14th, Junior Party the University of California, Berkeley; the University of Vienna; and Emmanuelle Charpentier; collectively, "CVC") filed its Substantive Motion No. 1 to be awarded priority benefit to their earlier priority applications: USSN 61/652,086, filed May 25, 2012 (P1); USSN 61/716,256, filed October 19, 2012 (P2); USSN 61/757,640, filed January 28, 2013 (P3); USSN 13/842,859, filed March 15, 2013; USSN 14/685,504, filed April 13, 2015; or USSN 15/138,604, filed April 26, 2016, in Interference No. 106,115 against Senior Party the Broad Institute, Harvard University, and the Massachusetts Institute of Technology.
On October 14th, Junior Party the University of California, Berkeley; the University of Vienna; and Emmanuelle Charpentier; collectively, "CVC") filed its Substantive Motion No. 1 to be awarded priority benefit to their earlier priority applications: USSN 61/652,086, filed May 25, 2012 (P1); USSN 61/716,256, filed October 19, 2012 (P2); USSN 61/757,640, filed January 28, 2013 (P3); USSN 13/842,859, filed March 15, 2013; USSN 14/685,504, filed April 13, 2015; or USSN 15/138,604, filed April 26, 2016, in Interference No. 106,115 against Senior Party the Broad Institute, Harvard University, and the Massachusetts Institute of Technology.CVC sets forth in detail the disclosure in its earlier priority applications for at least one embodiment falling within the scope of Count 1 of this interference. The brief recites the "ground-breaking" nature of their work, stating that the earliest priority document (P1) disclosed "the minimal components required to generate a functional CRISPR-Cas9 DNA-cleavage complex—Cas9, crRNA, and tracrRNA." In addition, CVC argues that this priority document "disclosed, for the first time, that complexes of Cas9 and a double- or single-molecule DNA-targeting RNA . . . are useful for targeted DNA cleavage and described numerous applications of this gene-editing technology, including modifying target DNA in eukaryotic cells," noting the many accolades bestowed upon CVC's inventors by the scientific community. CVC further argues that "[t]he CVC inventors immediately understood that the CRISPR-Cas9 DNA-cleavage complex could be used in a variety of different cellular and noncellular settings." The brief recites (prophetic) Example 1 in the P1 specification, and the (albeit prophetic) application of CRISPR technology to fish, human, and fruit fly cells.
The brief is careful to note that any failure of the P1 specification to show actual reduction to practice is not required to satisfy the requirement for entitlement benefit. CVC also cautions the Board against any attempt by the Broad to "erroneously to link the issues in this motion to the PTAB's termination of Interference No. 106,048 due to no interference-in-fact," stating that "the legal and factual issues raised here are fundamentally different from those decided in the prior '048 proceeding" based on the PTAB's own prior statements of the grounds for its no interference-in-fact determination. Rather, according to CVC:
[A person of ordinary skill in the art] reading P1 in light of the state of the art at the time of filing would have understood that the application describes and enables at least one embodiment within the scope of the count. Moreover, post-filing-date publications report successfully practicing CVC's claimed invention in eukaryotes using the very methods and components that P1 describes. The Board should therefore accord CVC the benefit of its first provisional application.
What follows is a succinct statement of the Precise Relief Requests (pursuant to PTAB rules) and support in the P1 specification for this relief, recited in the alternative with the other priority documents recited in CVC's request for relief. The standard, undisputed by the parties, is that to be accorded benefit of priority a prior application must show constructive reduction to practice (CRTP) regarding at least one embodiment falling within the scope of the count, citing Falkner v. Inglis, 448 F.3d 1357, 1362 (Fed. Cir. 2006). After setting out the legal grounds for CRTP, the brief then applies these rubrics to the disclosure in P1 for subject matter falling within the scope of the interference Count (which CVC argues satisfies these requirements). Along the way the brief also suggests that "Broad will doubtlessly rely on cherry-picked quotes about whether or not the inventors or experts knew CRISPR would work in eukaryotic cells before testing it," rejecting these anticipated arguments on the ground that CTRP is grounded on what is disclosed in the specification citing Ariad Pharms., Inc. v. Eli Lilly and Co., 598 F.3d 1336, 1351 (Fed. Cir. 2010) (en banc); Frazer v. Schlegel, F.3d 1283, 1288 (Fed. Cir. 2007); and Centrak, Inc. v. Sonitor Tech., Inc., 915 F.3d 1360, 1369 (Fed. Cir. 2019). The relevant P1 disclosure for CVC is that CRISPR is functional "when removed from its natural prokaryotic cellular milieu, which is highly relevant here because it establishes CVC's possession of the necessary and sufficient components for a functional CRISPR-Cas9 DNA-cleavage complex regardless of its environment" (emphasis in brief).
The argument is illustrated by Figures from the P1 application:
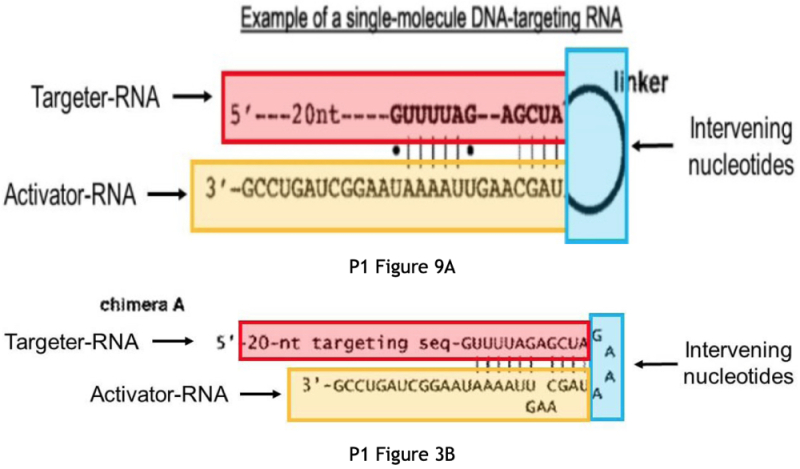
And explicit disclosure compared with the elements of the interference Count:
The brief then sets forth specific disclosure related to the elements of the Count (also provided in Appendix 3 entitled "Exemplary Evidence of Constructive Reduction to Practice of Count 1 in the P1 '068 Application"), and CVC further asserts that "[t]he inventors fully grasped the broad utility of such a method as aptly illustrated by the many types of 'target cells of interest' suitable for the methods described in P1, including a variety of eukaryotic cells (elements [1]-[2]) such as a fish, human, and fruit fly cell," noting that "[t]hese features are not merely recited in P1, but diagrammed, discussed, and specifically exemplified showing the inventors' possession" based on express disclosure cited with particularity in the brief (including specifically the prophetic use of CRISPR in fish cells). CVC presents explicit argument relating to what the skilled worker would understand CVC possessed and would be able to accomplish without undue experimentation with regard to the CRTP requirement for being accorded benefit. Finally, in this regard, the brief asserts that post-filing evidence (much of it by third parties) further supports a conclusion of constructive reduction to practice in the P1 disclosure:Within a year of CVC publishing that Cas9 and a [single guide RNA] such as chimera A (or a double-molecule RNA) formed a functional DNA-cleavage complex (Ex. 3202), other researchers used materially the same components and methods disclosed in P1 to practice the fish cell embodiment—objectively confirming that P1 enables making and using the fish cell of E1. While the studies described below published after P1's May 25, 2012 filing date, post-priority date evidence "may show, for example, that practicing the invention did not require undue experimentation," citing Amgen, Inc. v. Sanofi, 872 F.3d 1367, 1379 (Fed. Cir. 2017); In re Wands, 858 F.2d 731, 739 (Fed. Cir. 1988); In re Hogan, 559 F.2d 595, 605 (Fed. Cir. 1977).
CVC supports its allegations of what the skilled person would understand from the disclosure in the P1 specification by declaration testimony an expert, Dr. Peterson (who eventually will be subject to cross-examination by the Broad in this interference). Specifically, Dr. Peterson attests extensively to the applicability of CRISPR as set forth in P1 to use in human cells, which CVC asserts is also shown by third-party success in applying CRISPR to human cells, and fruit fly cells. Thus, CVC asserts "[i]n sum, the great weight of evidence compels a finding that P1 describes and enables each of the fish (E1), human (E2), and fruit fly (E3) cell methods, any one of which, on its own provides a CRTP of Count 1."
The brief then turns on the distinction between what was required for the Broad to prevail (as it did) in the earlier interference and in this one:
Broad's previously asserted arguments wrongly impose a reasonable expectation of success standard (obviousness) on §112, first paragraph, which contains no such standard. Obviousness "turns on . . . whether the claimed invention would have been obvious in view of the prior art." Allergan, Inc. v. Sandoz, Inc., 796 F.3d 1293, 1310 (Fed. Cir. 2015) (emphasis in original). "In contrast, the enablement inquiry turns on whether the skilled artisan, after reading the specification, would be able to make and use the claimed invention without undue experimentation." Id. (emphasis in original). Similarly, "[t]he standard for satisfying the written description requirement is whether the disclosure 'allow(s) one skilled in the art to visualize or recognize the identity of the subject matter purportedly described.'" Alcon, 745 F.3d at 1190 (emphasis added; citation omitted); see also, Ariad, 598 F.3d at 1351. Any argument to the contrary erroneously conflates obviousness with written description and enablement.
Finally, the brief argues in the alternative that the other applications CVC asserts for priority contain the P1 disclosure relied upon for priority in this brief, and thus for the same reasons (set forth in brief for each reference) CVC is entitled to priority to these applications, based on the "continuous chain" of priority in these CVC applications.
-
By Michael Borella —
 Berkheimer v. HP Inc. was decided by the Federal Circuit in February 2018 and stands for — in the words of Judge Moore of that Court — "the unremarkable proposition that whether a claim element or combination of elements would have been well-understood, routine, and conventional to a skilled artisan in the relevant field at a particular point in time is a question of fact." The Supreme Court's patent-eligibility opinions in Mayo Collaborative Srvs. v. Prometheus Labs, Inc. and Alice Corp. v. CLS Bank Int'l had not been perfectly clear on this issue, and the Federal Circuit appeared to have taken both sides at various times. Berkheimer settled the issue, for a while at least.
Berkheimer v. HP Inc. was decided by the Federal Circuit in February 2018 and stands for — in the words of Judge Moore of that Court — "the unremarkable proposition that whether a claim element or combination of elements would have been well-understood, routine, and conventional to a skilled artisan in the relevant field at a particular point in time is a question of fact." The Supreme Court's patent-eligibility opinions in Mayo Collaborative Srvs. v. Prometheus Labs, Inc. and Alice Corp. v. CLS Bank Int'l had not been perfectly clear on this issue, and the Federal Circuit appeared to have taken both sides at various times. Berkheimer settled the issue, for a while at least.HP petitioned the Supreme Court for certiorari in September 2018. In January of this year, the Court requested that the Solicitor General file a brief expressing the views of the United States in this matter. That brief finally arrived last week. In short, the Solicitor General is of the opinion that the overall lack of clarity regarding the fundamental issues surrounding patent-eligibility under 35 U.S.C. § 101 makes addressing the points of Berkheimer premature.
As background, the Supreme Court's Alice decision set forth a two-part test to determine whether claims are directed to patent-eligible subject matter under § 101. One must first decide whether the claim at hand is directed to a judicially-excluded law of nature, a natural phenomenon, or an abstract idea. If so, then one must further decide whether any element or combination of elements in the claim is sufficient to ensure that the claim amounts to significantly more than the judicial exclusion. But generic computer implementation of an otherwise abstract process does not qualify as "significantly more," nor will elements that are well-understood, routine, and conventional lift the claim over the § 101 hurdle.
While the Supreme Court did not specify whether the well-understood, routine, and conventional determination was to be carried out as a matter of law, fact, or a hybrid of both, its earlier Mayo Collaborative Srvs. v. Prometheus Labs, Inc. decision strongly suggested that the § 101 inquiry may overlap with fact-sensitive inquiries such as novelty under § 102 in at least some situations. However, the Federal circuit has stated, in OIP Techs., Inc. v. Amazon.com, Inc. that "[p]atent eligibility under 35 U.S.C. § 101 is an issue of law reviewed de novo." But in other cases, different panels have implied that facts may be considered.
This came to a head in Berkheimer, where Judge Moore wrote:
The question of whether a claim element or combination of elements is well-understood, routine and conventional to a skilled artisan in the relevant field is a question of fact. Any fact . . . that is pertinent to the invalidity conclusion must be proven by clear and convincing evidence. Like indefiniteness, enablement, or obviousness, whether a claim recites patent eligible subject matter is a question of law which may contain underlying facts. (Emphasis added.)
The outcome of the Court's factual analysis was that some of Berkheimer's dependent claims recite "limitations directed to the arguably unconventional inventive concept described in the specification" and therefore the case was remanded to the District Court for fact finding. Notably, Berkheimer did not win on the merits, but instead managed to get the District Court's invalidation of some of his claims reversed modulo further review.
In its petition, HP's question presented was "[w]hether patent-eligibility is a question of law for the court based on the scope of the claims or a question of fact for the jury based on the state of the art at the time of the patent." This question is somewhat deceptive, as the Federal Circuit explicitly stated that some patent-eligibility decisions can be made as a point of law, but others may require fact finding when the parties are in dispute over whether a claim element is well-understood, routine and conventional. HP's attempt to make this non-binary decision into a black-or-white issue does not reflect the nuances of the opinion.
Nonetheless, the Solicitor General begins his discussion with a blunt statement: "this Court's recent decisions have fostered uncertainty concerning those substantive Section 101 standards." This uncertainty lies mainly in the "the scope of the exceptions and the proper methodology for determining whether a particular patent implicates them."
Berkheimer being largely a procedural case would not be the appropriate vehicle for addressing the more fundamental substantive questions, in the Solicitor General's view. Indeed, "the allocation of decision-making authority between judge and jury . . . is deeply intertwined with the underlying legal standards that govern patent-eligibility." Thus, the Solicitor General opines that HP's question "would be difficult to answer in any cogent manner while uncertainty about the substance of the Section 101 inquiry persists." He further notes that Berkheimer's focus on step two of Alice is of limited use for purposes of clarifying § 101 since the uncertainty arises under both steps.
Indeed, any outcome of a Supreme Court ruling in Berkheimer could be rendered moot if the Court later rules on a more substantive § 101 case. The Solicitor General appears to be taking the position that there is little point to apply a Berkheimer band-aid when patent-eligibility actually needs major surgery.
The Solicitor General further observes that both parties are suggesting the Court use this case to address the larger § 101 issues. Yet, there is "significant disagreement about what the claimed invention actually comprises" in the disputed patent. The Solicitor General writes:
The district court appeared to view Berkheimer's patent as simply identifying features that it would be desirable for a software system to possess—one-to-many editing, increased efficiency, and reduced redundancy and storage costs—rather than as claiming a specific method for achieving those features and the corresponding benefits. In contrast, the court of appeals appeared to conclude that the claims at issue could be read as identifying a particular method that will achieve the desired results, and it declined to determine which view of the patent is correct in the case's current summary-judgment posture.
This implies that there is a potential problem with whether the patent conforms to the § 112(a) written description requirement. Particularly, the Solicitor General states that "[i]t would be difficult to provide meaningful clarity about Section 101's boundaries without first knowing whether and to what extent the invention represents a particular practical implementation of broader principles of software design to achieve the stated objective, or merely posits one possible way one might attempt to do so." In other words, if the scope of the invention is unclear, the Alice test is that much more difficult to apply.
The Solicitor General goes on to contend that this lack of clarity "may be exacerbated in the software-system context, in which this case arises." He even suggests that the Supreme Court is especially unequipped to address § 101 issues in software technologies, and that the software focus of Berkheimer would frustrate the Court's ability to clarify the larger principles underlying patent eligibility.
In accordance with these points, the Solicitor General concludes with the recommendation that "[r]ather than attempt to resolve broader questions of Section 101's substantive scope in the face of those difficulties, the Court should await an appropriate case that properly presents those broader questions without similar obstacles."
It should not be surprising, given the brashness of the Trump administration, that the executive branch would take two clear swipes at the judiciary: "You screwed up the law" and "you are not qualified to fix it." Despite the merits to both of these points, being a fly on the wall during the Court's conference on this case would likely prove interesting. But the important outcome here is that the government is arguing to let one of the very few patent-friendly § 101 decisions from the Federal Circuit stand.
