By Kevin E. Noonan —
 On October 14th, Senior Party the Broad Institute (joined by Harvard University and MIT) filed several authorized motions, including Substantive Motion No. 3 (to designate claims as not corresponding to the count), against Junior Party the University of California, Berkeley; the University of Vienna; and Emmanuelle Charpentier; collectively, "CVC." In its Motion No. 3, the Broad asks the Patent Trial and Appeal Board to designate some of its claims as not corresponding to the Count in Interference No. 106,115 as declared.
On October 14th, Senior Party the Broad Institute (joined by Harvard University and MIT) filed several authorized motions, including Substantive Motion No. 3 (to designate claims as not corresponding to the count), against Junior Party the University of California, Berkeley; the University of Vienna; and Emmanuelle Charpentier; collectively, "CVC." In its Motion No. 3, the Broad asks the Patent Trial and Appeal Board to designate some of its claims as not corresponding to the Count in Interference No. 106,115 as declared.
It should be appreciated that this Motion No. 3 is part and parcel of the Broad's motions to remake the contours of the interference, along with Motion No. 2 (to substitute the Count) and Motion No. 4 (to be awarded benefit of priority to USSN 61/736,527). In this Motion No. 3, the Broad reiterates the arguments made in Motion No. 2, that there are two embodiments of CRISPR, one involving single-molecule RNA guide RNA (which the Broad argues here is not recited in the claims it wants the Board to designate as not corresponding to the Count) and further that certain of the Broad's claims directed to "SaCas9" systems that require two or more NLSs do not correspond to the Count.
The brief parsed the Broad's claims into three categories of claims that do not correspond to the Count, depending on how the Board rules on Substantive Motions Nos. 1 and 2:
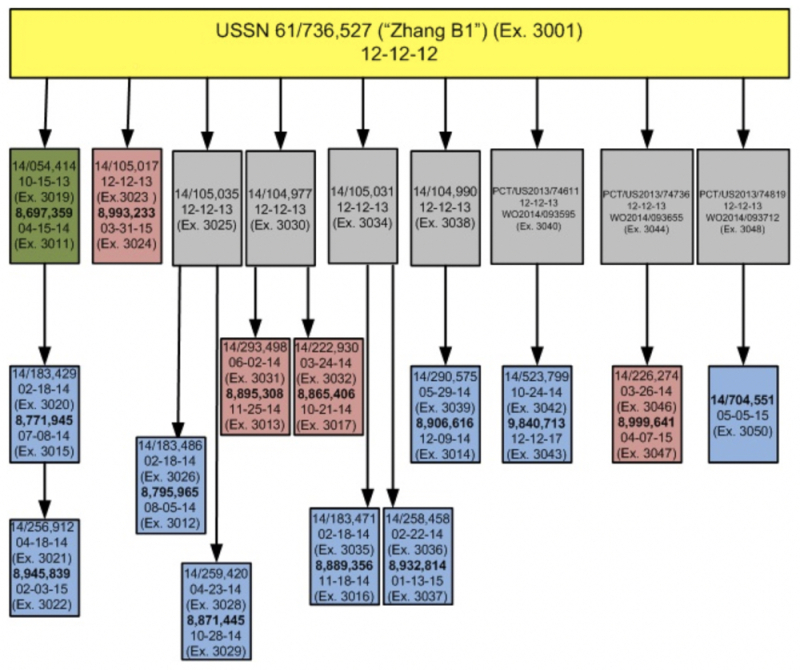
• USP 8,865,406 – Claims 1-30 (all); 8,871,445 – Claims 1-30 (all); USP 8,889,356 – Claims 1-30 (all); USP 8,932,814 – Claims 1-30 (all); USP 8,945,839 – Claims 1-28 (all); USP 8,993,233 – Claims 1-43 (all); USP 8,999,641 – Claims 1-28 (all); USP 8,697,359 – Claims 1-3, 5-10, 12-17, and 19-20; USP 8,771,945 – Claims 1-4 and 6-29; USP 8,895,308 – Claims 1-9 and 11-28; USP 8,906,616 – Claims 1, 3-4, 6-30; USP 9,840,713 – Claims 1-7, 10-15, 17-26, and 28-41; and U.S. Patent Application No. 14/704,551: in the event that the Board denies both Motions No. 1 and 2
• USP 8,865,406 – Claims 1-30 (all) and USP 8,895,308 – Claims 1-30 (all): in any event, claims reciting Ca9 from Staphylococcus aureus
• USP 8,871,445 – Claims 1-30 (all); USP 8,932,814 – Claims 1-30 (all); USP 8,993,233 – Claim 7; USSN 14/704,551 – Claims 9-11: clams reciting two or mote nuclear localization signal
And what would remain, should the Board grant this motion:
• U.S. Patent No. 8,697,359, claims 4, 11, and 18; U.S. Patent No. 8,795,965, claims 1-30 (all); U.S. Patent No. 8,771,945, claim 5; U.S. Patent No. 8,906,616, claims 2 and 5; and U.S. Patent No. 9,840,713, claims 8-9, 16, and 27
Regarding the first set of claims the Broad asserts do not correspond to Count No. 1 (which the Broad admits "includes the vast majority of [its] Involved Claims"), the Broad argues that the Count of the interference as declared is directed to "single-molecule guide RNA molecule"-comprising embodiments and the claims it has asked the Board to designate as not corresponding to the Count do not encompass these embodiments. The brief sets forth the Broad's understanding that, should the Board deny the Broad's Substantive Motions Nos. 1 and 2, then the interference will involve priority to such single-molecule guide RNA embodiments as a separate, patentable invention over claims that encompass both single-molecule and dual molecule embodiments. Thus, the Broad argues that its claims limited to dual-molecule embodiments do not correspond to the Count and the Board should so designate.
The brief cites for support for its motion CVC's statements (erroneously termed "Remarks provoking this Interference"; the interference was declared sua sponte by the Board not provoked by CVC) that CVC's generic claims "should not be included in an interference involving [its single-guide RNA claims], because those other claims are generic with respect to the guide RNA and are thus directed to a separately patentable invention." Under PTAB Rule 207(b), the Board provides relief to an inventor of a generic claim facing a specific count to move, as the Broad has done here, for their generic claims to be designated as not corresponding to the Count. The Broad asserts as reasons/justifications for de-designating certain (most) of its claims:
• Count 1 is limited to the single-molecule invention, and would not be determining priority to generic, non-limited RNA embodiments of CRISPR, "violating" the "'primary purpose of an interference'" to make a priority determination of "each of the common [patentably distinct] inventions claimed by the parties,'" citing Godtfredsen v. Banner, 598 F. 2d 589, 592 (C.C.P.A. 1979). The brief takes the opportunity to reiterate the unfairness of this outcome in view of the nature of their invention, e.g., a "breakthrough invention that revolutionized gene editing was the successful engineering of CRISPR-Cas9 systems for use in eukaryotic cells."
• There can be no interfering subject matter between the single-molecule RNA "invention" and generic, non-limited RNA CRISPR embodiments. Under these circumstances, the brief argues a two-count interference should be declared with each invention to reiterate its allegation that CVC had made a strategic decision not to present claims to eukaryotic embodiments of CRISPR in the earlier, '048 interference.
• It would be unfair to put all the Broad's claims at risk under these circumstances because it would "t[ie] its hand to its best generic RNA proofs," which the brief characterizes as being "particularly egregious given that Broad could have relied on its best proofs during the 048 Interference, when the Count properly reflected the scope of the parties' claims."
Next, the Broad argues that its claims to SaCas9 embodiments and to claims requiring two or more nuclear localization sequences (NLS's) do not correspond to Count 1. The Broad argues that these embodiments were not disclosed in the prior art and "provides a surprising combination of benefits not taught or suggested by the art" for both types of embodiments.
The brief then sets forth the Broad's characterization of the person of ordinary skill in the art, the properties and characterization of single- and dual-molecule RNA embodiments of CRISPR, a hagiography regarding the Broad's "pioneering eukaryotic CRISPR-Css9 work in 2011-2012" directed to dual-molecule RNA embodiments thereof, setting forth figures from contemporaneous grant proposals
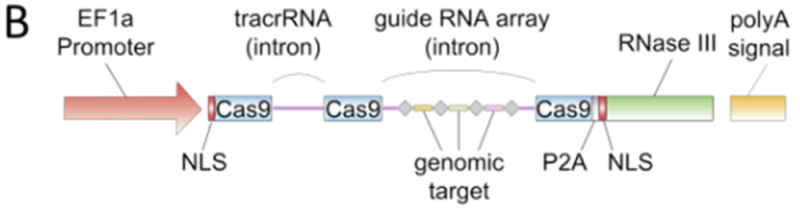
and scientific papers, and the "Zhang B1" provisional USSN 61/736,527

illustrating its reduction to practice of these embodiments of the invention.
The brief then reiterates the history of the '048 Interference, the PTAB's decision, and the Federal Circuit's affirmance of there being no interference-in-fact between the claims in that interference. Those claims were limited to eukaryotic cell embodiments for the Broad and unlimited with regard to target cell for CVC's claims; the brief notes that CVC provoked the claim to single-molecule CRISPR embodiments. Following these events, CVC pursued claims to "generic/non-limited, single-molecule, and dual-molecule RNA" CRISPR embodiments. This is followed by the Broad's continued inaccuracy in characterizing the interference as being "provoked" by CVC (in fact, the PTAB declared the interference sua sponte). The brief accurately cites CVC's arguments to the PTAB that single-molecule RNA CRISPR was patentably distinct from dual-molecule RNA embodiments.
Section V of the brief sets forth its arguments based on these facts, that certain of its claims should not correspond to Count 1. Citing 37 C.F.R. § 41.207 et seq., the brief asserts that the proper way to address the presumption regarding patentability must be addressed (as here) by filing a motion to designate claims as not corresponding to the count. And:
In the situation with a two-count interference – with a generic count and a separate species count – such a motion would be often be deferred until the priority phase and dismissed "unless there were a split award on priority, in which case proof that the generic invention antedates the priority proofs for the lost species count would likely justify relief."
But here the PTAB has not declared a two-count interference, and according to the brief, the first interference satisfied the question of priority to the "generic" invention (and once again CVC should not be heard to complain about the consequences of their strategic choices in the Broad's view).
The brief argues that the PTAB's failure to grant the motion "would conflict with the primary purpose of interferences" (awarding priority to the first to invent a patentable invention, wherein defining that patentable invention is the question before the Board as set forth in the Broad's motions in this interference). While the Count is directed to single-molecule RNA embodiments of CRISPR technology, the Broad's claims-in-interference encompass dual-molecule RNA embodiments as well (which the brief includes in its definition of the "generic, non-limited RNA invention"). The brief cites Godtfredsen (in which the court rejected a priority award to multiple species comprising a Markush group count in an instance where there were three patentably distinct inventions, because "the party who proved the earliest date of invention as to any one of the members of the group would be awarded priority as to the entire count") and Theeuwes v. Bogentoft, 2 U.S.P.Q.2d 1378 (B.P.A.I. 1986) (where there were two patentable inventions and the Board found it anomalous that party could be awarded priority a count covering species A and B while proving priority to species B alone) in support of its argument, and cites Ex Parte Hardman, 142 U.S.P.Q. (BNA) ¶ 329 (July 28, 1964), as an instance where the Board held a genus claim unpatentable over a species disclaimed in an earlier interference (finding it inequitable to find otherwise). The brief ties this precedent to the claims in this interference it had moved to dedesignate, on the grounds that "Dr. Zhang succeeded with both dual and single molecule RNA systems" and "[t]hus, it would be entirely inappropriate to award priority to the broad invention of eukaryotic CRISPR systems to a party who made a species invention long after the break." The remainder of the Broad's argument in this regard expound on the purported unfairness in determining priority to the claims it had moved to dedesignate based on Count 1.
The brief then supplements with greater specificity the distinctions between "generic, non-limited RNA" embodiments of CRISPR claimed in the claims the Broad moves to dedesignate and the single-molecule RNA embodiments encompassed in the Count, particularly with regard to definitions for "guide RNA," tracr RNA," and other species comprising various permutations of CRISPR technology, in contrast to CVC's interpretation of these terms in the claims.
Finally, the brief reiterates the Broad's arguments regarding claims reciting SaCas9 and species comprising more than 2 NLS's as not corresponding to the Count. For the former species, the brief asserts that:
As of 2012, there were more than 600 bacterial Cas9 orthologs. MF 29; Ex. 3226, Ran et al., In vivo genome editing using Staphylococcus aureus Cas9, 520 Nature 186-191 (2015), 1; Ex. 3401, Seeger ¶¶ 6.5, 6.13. Broad is unaware of any prior art suggesting the use of, or providing reasons for the selection of, SaCas9 in a CRISPR system adapted for use in eukaryotes. Indeed, CVC, in its Opposition to a similar motion in the 048 Interference, did not point to any such prior art either. Ex. 3115, Paper 689 (Opp. to 048 Mot. 5). Where, as here, the prior art choices are vast, the prior art must provide some reason to make the selection in order to find obviousness. Ortho-McNeil Pharm., Inc. v. Mylan Labs., Inc., 520 F. 3d 1358, 1364 (Fed. Cir. 2008). Here, the prior art fails to provide, or even suggest, a reason to select SaCas9.
The brief similarly argues that the art did not suggest embodiments comprising more than two NLSs in CRISPR systems, and that its claims directed to such embodiments should be found not to correspond to the Count on that basis:
The prior art lacked disclosure of advantages for two or more NLS(s) demonstrates that the use of two or more NLSs and there is no teaching, suggestion, motivation or reason in the prior art that would have caused a POSA to use two or more NLSs. . . . Therefore, claims including a limitation requiring two or more NLSs should be designated as not corresponding to Count 1.
 Last month, the United States government, acting on behalf of its Department of Health and Human Services (HHS), filed suit in Delaware against Gilead Sciences, Inc. and Gilead Sciences Ireland UC for infringing four patents covering inventions developed by scientists at the Centers for Disease Control and Prevention. The patents all cover methods for a type of medical regimen known as "pre-exposure prophylaxis" (PrEP) for the prevention of HIV. More specifically, each of the four patents' claims recite:
Last month, the United States government, acting on behalf of its Department of Health and Human Services (HHS), filed suit in Delaware against Gilead Sciences, Inc. and Gilead Sciences Ireland UC for infringing four patents covering inventions developed by scientists at the Centers for Disease Control and Prevention. The patents all cover methods for a type of medical regimen known as "pre-exposure prophylaxis" (PrEP) for the prevention of HIV. More specifically, each of the four patents' claims recite: The lawsuit is highly noteworthy for many reasons. It is very unusual — not unheard of, but very unusual — for the U.S. government, acting as a patent owner, to sue a company for patent infringement. Second, the Complaint is extremely detailed in explaining the history of events leading to the invention as well as the history of interaction between HHS and Gilead leading to the filing of the Complaint. Third, it does not take much reading between the lines to see that one of the government's goals is to make the PrEP regimen as readily available and cost effective in the U.S. as it already is in many European and other countries, with Gilead being alleged as a primary roadblock in that path.
The lawsuit is highly noteworthy for many reasons. It is very unusual — not unheard of, but very unusual — for the U.S. government, acting as a patent owner, to sue a company for patent infringement. Second, the Complaint is extremely detailed in explaining the history of events leading to the invention as well as the history of interaction between HHS and Gilead leading to the filing of the Complaint. Third, it does not take much reading between the lines to see that one of the government's goals is to make the PrEP regimen as readily available and cost effective in the U.S. as it already is in many European and other countries, with Gilead being alleged as a primary roadblock in that path.