By Kevin E. Noonan —
 As the COVID-19 pandemic has disrupted life throughout the world this spring, bats have been a prominent feature in news stories and recriminations about how the pandemic started (and being blamed even more than happenings in China, although to be fair the effects of the pandemic, good and bad, have been more the consequence of how countries and their citizens and their leaders have reacted to the virus than merely the virus itself). In view of the state of modern biology, it is not surprising that the bat genome is an interesting area of study, and this week an international team of researchers published results that shed light on bat biology and evolution (if not on their propensity to harbor viruses capable of such devastating effects on humans).
As the COVID-19 pandemic has disrupted life throughout the world this spring, bats have been a prominent feature in news stories and recriminations about how the pandemic started (and being blamed even more than happenings in China, although to be fair the effects of the pandemic, good and bad, have been more the consequence of how countries and their citizens and their leaders have reacted to the virus than merely the virus itself). In view of the state of modern biology, it is not surprising that the bat genome is an interesting area of study, and this week an international team of researchers published results that shed light on bat biology and evolution (if not on their propensity to harbor viruses capable of such devastating effects on humans).
The paper, entitled "Six reference-quality genomes reveal evolution of bat adaptations," was published in Nature. As the authors explain, the paper provides "the first reference-quality genomic sequence of six bat species, specifically Rhinolophus ferrumequinum, Rousettus aegyptiacus, Phyllostomus discolor, Myotis myotis, Pipistrellus kuhlii, and Molossus molossus (only a few of the 1,400 known bat species, comprising 20% of all existing mammalian species). This genomic information allowed these scientists to elucidate bat evolutionary antecedents, specifically their origin within the Scrotifera, a broad group that encompasses most placental mammals. Not surprisingly, these studies also revealed positive selection for hearing-related genes, and selective loss of certain immunity-related genes, in particular ones (such as NF-κB regulators) related to inflammation. There is a striking expansion of antiviral APOBEC3 genes the authors believe is related to the bat's exceptional immunity to viral infection.
The researchers had a number of unique phenotypic traits exhibited by bats to explore for their genetic bases, including flight, echolocation, extreme longevity, and perhaps more relevant to their current notoriety, unique immunity. Applying what they termed "state-of-the-art sequencing technologies and assembly algorithms," these scientists report that they were able to generate contiguous genomic sequences that were ~100-fold more contiguous than earlier attempts (and thus less prone to error) and at ~100% completeness. From these data, the authors report they annotated between 19,122 and 21,303 protein-coding genes with an annotation quality higher than that obtained to date for dog, cat, horse, cow and pig, and surpassed (not surprisingly) only by mouse and human annotated genomic sequences. The size of the bat genome determined using these analyses is approximately 2 gigabasepairs (Gb), which the researchers characterize as being smaller than for other placental mammals (which range from 2.5-3.5Gbp). This disparity was found to be related to lower content of transposable elements in the bat genome, with there being extreme variability (in both type and number) of more recently inserted transposable elements compared with other mammals.
Evolutionarily, the genomic sequence data is consistent with one of four clades that diverged in the late Cretaceous (100-66 million years ago, Mya), illustrated by this diagram (click to expand):
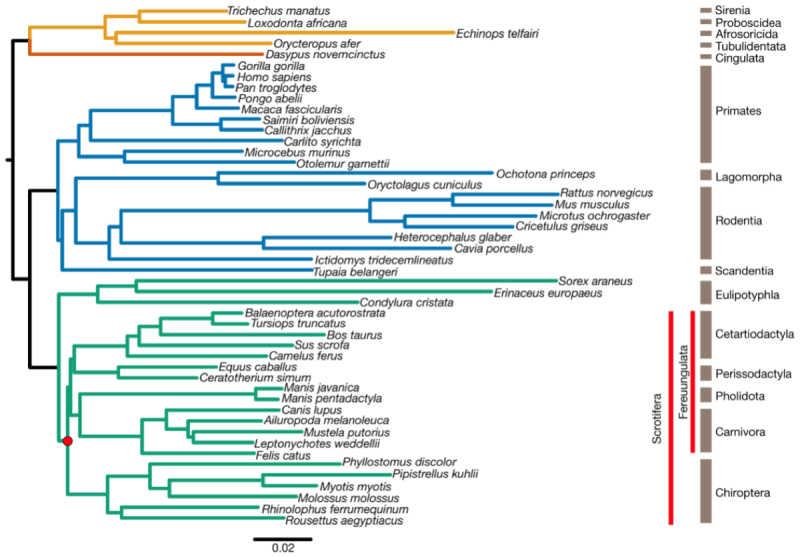 (where green designates the Laurasitheria which contain the origins of bats; blue, Euarchontoglires; orange, Xenarthra; and yellow, Afrotheria). These results arose from comparisons of 12,931 orthologous protein-coding genes and 10,857 orthologous conserved noncoding elements from genomes of 48 mammalian species.
(where green designates the Laurasitheria which contain the origins of bats; blue, Euarchontoglires; orange, Xenarthra; and yellow, Afrotheria). These results arose from comparisons of 12,931 orthologous protein-coding genes and 10,857 orthologous conserved noncoding elements from genomes of 48 mammalian species.
Having developed these phylogenetic relationships, the authors next turned to what they termed "the genomic basis of exceptional traits" that have arisen in bats. These comparisons identified nine genes having undergone positive selection from ancestral species, including LRP2, TJP2, and SERPINB6 genes, all of which are involved, inter alia, in hearing. Specifically, these genes are expressed in cochlea and, in humans are associated with deafness. Interestingly, the LRP2 gene contains a sequence change that encodes an amino acid substitution (methionine) specific to bats capable of echolocation; bats that do not express this phenotype — such as pteropodid bats — have a different amino acid (threonine) encoded at this position. Patterns of inheritance of microduplications in the TJP2 gene were interpreted by these scientists to mean that the echolocation-related mutations arose in bat ancestors but were lost in bats who do not exhibit this trait, providing an evolutionary answer to the question of whether echolocation had arisen multiple times independently in these bats.
The comparative screening experiments also provided information on genes related to immunity. These included "the B-cell-specific chemokine CXCL13, the asthma-associated NPSR1 and INAVA (a gene that is involved in intestinal barrier integrity and enhancing NF-κB signaling in macrophages). Ten additional immunity-related genes identified in these studies included "IL17D and IL1B (which are involved in immune system regulation and NF-κB activation), and LCN2 and GP2 (which are involved in responses to pathogens)." These genes showed the presence of "bat-specific" sequence variants that had undergone positive selection during bat evolution, supporting their role in the development of the pathogen-resistance phenotype seen in bats. In addition to these bat-specific sequence variants, the paper reports ten genes lost or inactivated in bats that are expressed in non-bat members of the Laurasiatheria. These include two genes, LRRC70 (known to "potentiates cellular responses to multiple cytokines and amplif[y] NF-κB activation mediated by bacterial lipopolysaccharides") and IL36G (that encodes a pro-inflammatory interleukin).
Another group of immune-related genes are those that have been differentially amplified in bats; most relevant are genes in the APOBEC gene family which is expanded in bats at the APOBEC3 locus (which encodes "DNA- and RNA-editing enzymes that can be induced by interferon signaling and are implicated in restricting viral infection and transposon activity"). These genes were found as a "small" expansion in ancestral bat species and "multiple, lineage-specific expansions that involve up to 14 duplication events" in later-arising bat species. Consistent with the evolution of pathogen (particularly viral) resistance in bats, the genomic analyses reported in this paper found evidence of prior viral infection in the form of endogenous viral elements (EVEs) in three predominant non-retroviral forms. These genes encoded viral envelope (env) proteins similar to alpha retroviruses which had previously been thought to be confined to avian species.
The paper also reports evolution-related changes in non-coding RNAs. These results showed that "nearly all of the annotated noncoding RNA genes are shared across all six bat genomes . . . , and between bats and other mammals [for example, 95.8–97.4% are shared between bats and humans]." Of the 286 known conserved miRNA gene families found in mammals, eleven were "significantly contracted" in bats and thirteen were lost. For single-copy miRNA genes, one (known as miR-337-3p) had unique variation in bats related to binding specificity and was "pervasively expressed." The researchers also reported finding 122-261 novel miRNAs, with twelve being shared in all six bat species interrogated (two of which were shown to be functional in regulating gene expression).
The authors conclude by stating:
Our conservative genome-wide screens investigating gene gain, loss and selection revealed novel candidate genes that are likely to contribute tolerance to viral infections among bats. Consistent with this finding, we also found that bat genomes contain a high diversity of endogenized viruses. We also uncovered genes involved in hearing that exhibit mutations specific to laryngeal-echolocating bats and ancestral patterns of selection. . . . Finally, we identified and experimentally validated miRNAs that are evolutionary novelties or that carry bat-specific changes in their seed sequence. Changes in these important regulators of gene expression may have contributed to changes in developmental and behavioural processes in bats.
And perhaps more importantly these researchers recognized that:
These high-quality bat genomes, together with future genomes, will provide a rich resource to address the evolutionary history and genomic basis of bat adaptations and biology[]. These genomes enable a better understanding of the molecular mechanisms that underlie the exceptional immunity and longevity of bats, allowing us to identify and validate molecular targets that ultimately could be harnessed to alleviate human ageing and disease. For example, we predict that our reference-quality bat genomes will be tools that are heavily relied upon in future studies focusing on how bats tolerate coronavirus infections. This is of particular global relevance given the current pandemic of coronavirus disease 2019 (COVID-19), and ultimately may provide solutions to increase human survivability—thus providing a better outcome for this, and future, pandemics.
* The researchers hie from the Max Planck Institute of Molecular Biology and Genetics, Institute of Systems Biology, and Institute of Physics, Dresden, DE; The Max Planck Institute for Psycholinguistics, NL; the School of Biology and Environmental Science, the Conway Institute of Biomolecular and Biomedical Science, and the Earth Institute, University College Dublin; the Australian National University, Canberra; the Department of Biological Science, Texas Tech; the Department of Ecology and Evolution, and Consortium for Inter-disciplinary Environmental Research, SUNY Stony Brook; Department of Sciences, John jay College of Criminal Justice; Department of Biological Sciences, University of Bristol; Department of Biology, University of Konstanz; Smithsonian Tropical Research Institute, Panama City; ISEM, University of Montpellier; Zoological Institute and Museum, University of Greifswald; Department of Migration, Max Planck Institute of Animal Behavior, Radolfzell; Vertebrate Genomes Laboratory and the Laboratory of Neurogenetics of Language, The Rockefeller University; Howard Hughes Medical Institute, Chevy Chase, Maryland; Donders Institute for Brain, Cognition and Behavior, NL; and the Facility of Computer Science, Technical University, Dresden










