By Kevin E. Noonan —
In the latest development in Interference No. 106,115 between Senior Party The Broad Institute, Harvard University, and the Massachusetts Institute of Technology (collectively, "Broad") and Junior Party The University of California/Berkeley, the University of Vienna, and Emmanuelle Charpentier (collectively, "CVC"), Broad filed its priority motion (which, as Senior Party they were not obliged to do).
 Broad's brief is a hybrid: on the one hand establishing its dates of conception and actual reduction to practice, and on the other denigrating CVC's claims of conception and reduction to practice. The latter portion of its argument hinges on two concepts: first, that eukaryotic applications of CRISPR-Cas9 are governed by the principle of "simultaneous conception and reduction to practice" (a contention for which Broad has the burden of convincing the Patent Trial and Appeal Board), and second, that CVC's admissions prevent them from successfully arguing (as they do in their priority brief) conception followed by acting with diligence to reduce their invention to practice; this contention includes "admission by omission" to the extent, inter alia, that CVC neglected to include their experiments using fish cells from the provisional application, No. 61/716,256, filed October 19, 2012).
Broad's brief is a hybrid: on the one hand establishing its dates of conception and actual reduction to practice, and on the other denigrating CVC's claims of conception and reduction to practice. The latter portion of its argument hinges on two concepts: first, that eukaryotic applications of CRISPR-Cas9 are governed by the principle of "simultaneous conception and reduction to practice" (a contention for which Broad has the burden of convincing the Patent Trial and Appeal Board), and second, that CVC's admissions prevent them from successfully arguing (as they do in their priority brief) conception followed by acting with diligence to reduce their invention to practice; this contention includes "admission by omission" to the extent, inter alia, that CVC neglected to include their experiments using fish cells from the provisional application, No. 61/716,256, filed October 19, 2012).
The comparison between the two inventive timelines asserted by Broad was set forth in the motion by this graphic:
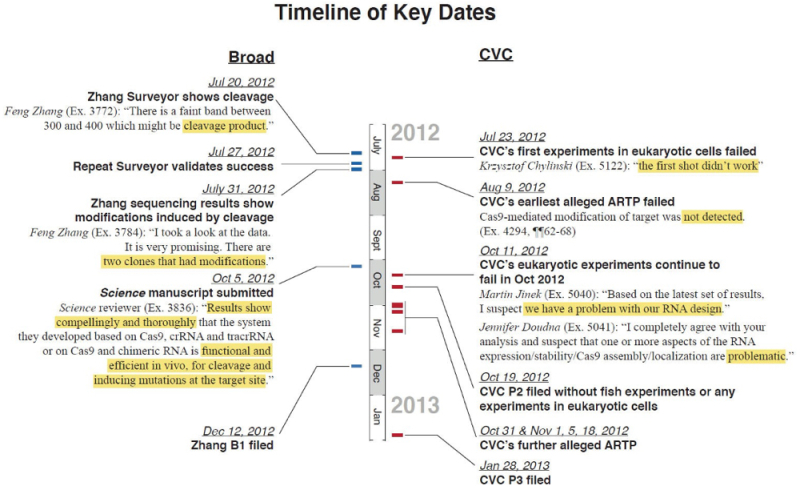
Broad's brief is explicit, saying "CVC is not entitled to any of its asserted conceptions, all of which allegedly occurred before CVC had any success in eukaryotic cells. Count 1 specifically requires specific biologic results in a eukaryotic cell for which the CVC inventors could have had no reasonable expectation of success absent successful eukaryotic experiments and thus could have no conception," citing Hitzeman v. Rutter, 243 F.3d 1345, 1358 (Fed. Cir. 2001), and the "law of the case," no doubt a reference to the outcome of the earlier Interference between the parties (No.106,048). As illustrated in the timeline, Broad asserts an actual reduction to practice on either July 20th or 27th (depending on when Broad's inventors can be found to have "recognized and appreciated [their] success"). These experiments and their results were corroborated, according to the brief, by two witnesses (Dr. Andrew Ellington and Dr. Ronald Breaker) as well as by contemporaneous communications by Dr. Zhang and declarations of other laboratory members (whose status as inventors vel non is in dispute; see "CVC Files Substantive Motion No. 3 (for improper inventorship) and Broad Opposes"). Broad's brief also cites multiple other instances of actual reduction to practice (as illustrated in the timeline). In contrast, Broad argues that "CVC did not achieve any success until, at the absolute earliest, October 31, 2012" based on the statements synopsized in the illustration for all earlier actual reduction to practice dates asserted by CVC.
In addition, Broad argues that, while not needed, the Board can also consider evidence of Dr. Zhang's earlier conception beginning on June 26, 2012 (although the arguments asserted for this date are accompanied by the same language CVC used for its conceptions, such as "[h]e further reasonably expected" [that his CRISPR system would work in eukaryotic cells], and include assertions of diligence from that time to the asserted actual reduction to practice in July 2012). Should the Board still consider CVC's evidence for earlier date(s) of conception, Broad's motion argues that "Dr. Zhang's successful eukaryotic experiments with his dual-molecule RNA CRISPR-Cas9 systems, which occurred well before all of CVC's alleged conceptions, should be taken into account"; Broad's relevant insight being that "[t]hese experiments answered the key question of whether a Cas9 system could be engineered to overcome the expected hurdles in eukaryotic cells," and conceding that "the concept of linking already-hybridized RNA using known linking methods would have been routine" (and thus distinguishing Broad's work and arguments about the routine status of reducing CRISPR to eukaryotic cell environment asserted by CVC in their priority motion).
These arguments are supported by several appendices, including a chart showing Broad diligence as Appendix C.
Turning to the legal argument, Broad leads with its argument regarding the "law of the case": "mere words and in vitro experiments do not provide a reasonable expectation of success in achieving that specific biological result (a functional eukaryotic CRISPR-Cas9 system)." Properly asserting that "applicable Federal Circuit precedent is clear that to have conception of a count claiming a specific biological result, the inventor must first have a reasonable expectation of success that the claimed result will be achieved," the brief goes on to assert that any "reasonable expectation of success that the claimed result will be achieved" requires that the claimed result actually be achieved. The motion argument cites in support the situation in Hitzeman v. Rutter, where a party's claimed conception to a count directed to recombinant production of hepatitis B surface antigen (HBsAg) in yeast failed because "[o]ne skilled in the art at the time this application was filed would not have been able to reasonably predict that HBsAg could be expressed by yeast [and done so in particle form]." Id. at 1357 (emphasis added). Thus is the parallel established: according to Broad, the uncertainty in practicing CRISPR in a eukaryotic cell (which was convincing to the Board in deciding the earlier interference between these parties) precludes CVC from depending on its earlier dates of conception, and the Board should recognize Broad's earlier dates of reduction to practice and award priority on that basis.
The brief includes a certain degree of hagiography regarding Dr. Zhang having at least questionable relevance (but understandable in light of CVC's similar citation of the Novel Prize award to their inventors, Jennifer Doudna and Emmanuelle Charpentier). Much of this history (including a grant proposal submitted in January 2012) certainly indicates Dr. Zhang's early recognition of the possible usefulness of CRISPR in eukaryotic cells and experiments to achieve this result, but none of it is asserted as part of Broad's conception (although citation of these early efforts (prior to April 2012) support Broad's attempts to put their inventors on the same chronological playing field as CVC's inventors). The brief then tells the story of Dr. Marraffini's contribution (stemming from information he "heard about at a public conference") regarding sg-RNA CRISPR embodiments, establishing the basis for Broad's contention that this date, June 26, 2012 was Dr. Zhang's first conception of an invention corresponding to the Count in the interference) (the brief is also careful to assert that "[b]ecause Dr. Zhang had already engineered CRISPR-Cas9 systems with the analogous dual-molecule RNA CRISPR-Cas9 system to function in eukaryotic cells, he expected on June 26, 2012 when he received the email from Dr. Marraffini that he could successfully implement his engineered CRISPR-hSpCas9 system with a chimeric RNA having a sufficient tracrRNA length for use in eukaryotic cells in a manner analogous to his dual-molecule RNA system"). Broad's motion also seeks to establish that Dr. Zhang (in contrast to CVC's inventors) had a reasoned basis for expecting that "a CRISPR-Cas9 system could be engineered to overcome the major obstacles presented by eukaryotic cells including, for example, chromatin, RNA degradation, toxicity, and successful co-localization in the eukaryotic cellular milieu," this being his earlier successes in practicing the three-molecule variation of CRISPR in eukaryotic cells. This for Broad establishes conception, wherein "Dr. Zhang had a definite and permanent idea of the complete operative invention of Count 1."
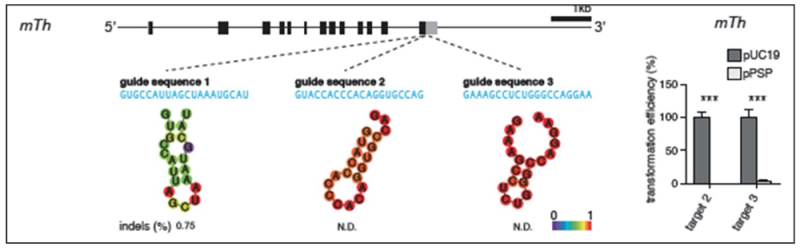
Like in CVC's brief, Broad's motion includes an illustration of the experimental design of the sg-RNA used in these experiments, as described in an e-mail to Dr. Le Cong:

Followed by a description of Dr. Zhang's asserted successful reduction to practice of embodiments of sg-RNA CRISPR in eukaryotic mouse cells on July 20th and 26th. This success was verified according to the motion on July 31, 2012, showing by comparative sequencing the expected deletion in the target sequence:

These data and experimental results are supported as they must be by collaborative declarations by several non-inventors (at least until the PTAB renders a decision on CVC's misjoinder of inventorship motion). And with regard to the requirement that an inventor recognize her invention the motion asserts communications from Dr. Zhang as well as the submission of a manuscript to Science on October 5, 2012 containing in a Figure a map of the vector used in the July 2012 experiments:
The motion also asserts multiple additional actual reduction to practice events in August 2012. These and other experiments were contained, according to Broad, in the "landmark" January 3, 2013 Science publication by Cong et al.
Broad's arguments then provide the Board with a detailed description of the correspondence between its experimental evidence for actual reduction to practice and claim 18 of Broad U.S. Patent No. 8,697,359 or claim 156 of CVC U.S. Patent Application No. 15/981,807, extensively corroborated by non-inventor declarants for experiments conducted in July, August, and September 2012 and the October 5, 2012 Science manuscript. This Section also includes positive statements about the authors' achievements by Science reviewers (which of course have no legal effect except in support of Broad's assertions of how these experiments would be understood by those of at least ordinary skill in the art). And the motion again illustrates how Dr. Zhang "recognized and appreciated" his achievement regarding eukaryotic embodiments of CRISPR, from contemporaneous, corroborated statements.
The motion provides further argument regarding earlier conception (June 26, 2012) by Dr. Zhang than the CVC inventors, based on his ability to have a reasonable expectation of success based on earlier successful experiments, something Broad asserts CVC did not have based on experimental failures and negative statements first adduced in the '048 Interference. The motion also argues that, should the PTAB give credence to CVC's asserted conception date of March 2012 then Dr. Zhang's work prior to March 2012 should also be considered (although any such consideration would need to also take into account Broad's disclosure that sg-RNA embodiments were first considered after the June 26, 2012 communication from Dr. Marraffini). And these arguments track some of the same resort to "routine techniques" asserted by CVC in support of its priority position.
The motion concludes with assertions of diligence, supported by a log of day-to-day diligence provided in Appendix C of the motion, and that Dr. Zhang did not abandon, suppress, or conceal Broad's invention, based inter alia on the October 5, 2012 Science manuscript and Broad's provisional application filing on December 12, 2012 (which the Board recognized as Broad's earliest priority document; see "PTAB Grants Broad Motion No. 4 for Priority Benefit to U.S. Provisional Application No. 61/736,527").
From all these facts Broad argued it is entitled to priority award in this Interference.
* As a reminder, in an interference, the Junior Party (CVC) bears the burden of showing actual reduction to practice before reduction to practice by the Senior Party (Broad). Cabilly v. Boss, 55 USPQ2d 1238, 1254-5 (BPAI 2000), citing Boises v. Benedict, 27 F.3d 539, 541-542, 30 USPQ2d 1862, 1864 (Fed. Cir. 1994); 37 C.F.R. §41.121. This showing must be by a preponderance of the evidence. Hahn v. Wong, 892 F.2d 1028, 1032, 13 USPQ2d 1313, 1317 (Fed. Cir. 1989), citing Oka v. Youssefyeh, 849 F.2d 581, 7 USPQ2d 1169, 1171 (Fed. Cir. 1988).
The inventor is the party that first reduced the invention to practice, unless the other party can show that they were the first to conceive the invention and exercised reasonable diligence from a time just prior to the other party's conception through their own reduction to practice. Hitzeman v. Rutter, 243 F.3d 1345, 1353, 58 USPQ2d 1161, 1166 (Fed. Cir. 2001), citing Cooper v. Goldfarb, 154 F.3d 1321, 1327, 47 USPQ2d 1896, 1900-01 (Fed. Cir. 1998). A junior party that shows that she was first to reduce the invention to practice prevails, Hahn v. Wong 892 F.2d at 1317, and a junior party has no obligation to establish diligence in these circumstances, since it is first actual reduction to practice, not the relative diligence of the parties, that carries the junior party's priority burden. Steinberg v. Seitz, 517 F.2d 1359, 1364, 186 USPQ 209, 213 (CCPA 1975).
Conception is "the formation in the mind of the inventor, of a definite and permanent idea of the complete and operative invention, as it is hereafter to be applied in practice." Hybritech Inc. v. Monoclonal Antibodies, Inc., 802 F.2d 1367, 1376, 231 U.S.P.Q. 81, 87 (Fed. Cir. 1986), citing Coleman v. Dines, 754 F.2d 353, 359, 224 USPQ 857, 862 (Fed. Cir. 1985). Since conception occurs in the mind of the inventor, there must be "corroborating evidence of a contemporaneous disclosure that would enable one of ordinary skill to make the invention." Burroughs Wellcome, Id. at 1919, citing Coleman v. Dines, 754 F.2d 353, 359, 224 USPQ 857, 862 (Fed. Cir. 1985). However, conception of a method does not require knowledge that the invention will work for its intended purpose. Burroughs Wellcome Co. v. Barr Labs, Inc., 40 F.3d 1223, 32 USPQ 2d 1915 (Fed. Cir. 1994). Relevant to Broad's arguments in their motion, in Burroughs Wellcome, the claims of the patents-in-suit were directed to methods for using AZT for treating AIDS, and the issue was whether the AZT inventors had conceived of the claimed methods before obtaining evidence that AZT could indeed provide an effective treatment for HIV infection. Id. at 1225. The Burroughs Wellcome defendants argued that for an invention in an "uncertain or experimental discipline, where the inventor cannot reasonably believe an idea will be operable until some result supports that conclusion," conception occurs only when there is experimental confirmation that the invention works for its intended purpose. Id. at 1228. The Federal Circuit was clear, stating: "[b]ut this is not the law. An inventor's belief that his invention will work or his reasons for choosing a particular approach are irrelevant to conception." Id., citing MacMillan v. Moffett, 432 F.2d 1237, 1239, 167 U.S.P.Q. 550, 552 (CCPA 1970). This is sufficient for conception, unless there is evidence of subsequent experimental failure (the argument Broad relies upon in their argument against CVC): "[a] conception is not complete if the subsequent course of experimentation, especially experimental failures, reveals uncertainty that so undermines the specificity of the inventor's idea that it is not yet a definite and permanent reflection of the complete invention as it will be used in practice." Id. at 1229, citing Rey-Bellet v. Engelhardt, 493 F.2d 1380, 1387, 181 U.S.P.Q. 453, 457-58 (CCPA 1974).
Unlike conception, actual reduction to practice in the chemical or biotechnology arts requires that the inventor has reduced to practice an embodiment of the compound that works for its intended purpose. Cooper v. Goldfarb, 154 F.3d 1321, 1327, 47 USPQ2d 1896, 1901 (Fed. Cir. 1998). More than the existence of laboratory notebook pages showing an embodiment within the scope of the count must be corroborated for reduction to practice; corroboration that the embodiment was actually made is needed. Hahn v. Wong, 892 F.2d 1028, 1032, 13 USPQ2d 1313, 1317 (Fed. Cir. 1989). If the embodiment requires testing to establish its usefulness for its intended purpose, corroborated evidence of such testing is required. Newkirk v. Lulejian, 825 F.2d 1581, 1582, 3 USPQ2d 1793, 1794 (Fed. Cir. 1987). When testing is required to determine whether an embodiment of an invention works for its intended purpose, whether a party can sustain its burden of showing actual reduction to practice by a preponderance of the evidence depends on the extent, nature and results of the testing required. Scott v. Finney, 34 F.3d 1058, 1061, 32 USPQ2d 1115, 1117-8 (Fed. Cir. 1994) , citing Newkirk v. Lulejian, 825 F.2d 1581, 1582, 3 U.S.P.Q.2d 1793, 1794 (Fed. Cir. 1987).
 Motion practice continues in Interference No. 106,115 between Senior Party The Broad Institute, Harvard University, and the Massachusetts Institute of Technology (collectively, "Broad") and Junior Party the University of California/Berkeley, the University of Vienna, and Emmanuelle Charpentier (collectively, "CVC"), with CVC filing on January 6th its Reply to Broad's opposition to CVC's Miscellaneous Motion No. 6 for leave to subpoena discovery (including depositions) from Luciano Marraffini and Shuailiang Lin, neither of whom is a party to this interference (pursuant to an Order authorizing filing of this Reply issued by the Board on December 28th).
Motion practice continues in Interference No. 106,115 between Senior Party The Broad Institute, Harvard University, and the Massachusetts Institute of Technology (collectively, "Broad") and Junior Party the University of California/Berkeley, the University of Vienna, and Emmanuelle Charpentier (collectively, "CVC"), with CVC filing on January 6th its Reply to Broad's opposition to CVC's Miscellaneous Motion No. 6 for leave to subpoena discovery (including depositions) from Luciano Marraffini and Shuailiang Lin, neither of whom is a party to this interference (pursuant to an Order authorizing filing of this Reply issued by the Board on December 28th). Broad in its opposition contended that any such evidence would be redundant over evidence available from other witnesses and documents, and that much of any such testimony was not relevant to Broad's priority proofs. Broad further argued that there were no inconsistencies between whatever testimony CVC could adduce from these witnesses and the "allegations" in its priority statement and evidence it would proffer with its (at the time, to be filed) Motion for Priority.
Broad in its opposition contended that any such evidence would be redundant over evidence available from other witnesses and documents, and that much of any such testimony was not relevant to Broad's priority proofs. Broad further argued that there were no inconsistencies between whatever testimony CVC could adduce from these witnesses and the "allegations" in its priority statement and evidence it would proffer with its (at the time, to be filed) Motion for Priority.