By Kevin E. Noonan —
Senior Party Toolgen and Junior Parties The Broad Institute, Massachusetts Institute of Technology, and Harvard University (collectively, "Broad") in Interference No. 106,126 and University of California/Berkeley, the University of Vienna, and Emmanuelle Charpentier (collectively, "CVC") in Interference No. 106,127 each filed Lists of Proposed Motions that the Board considered recently and responsive thereto will issue its rulings shortly (see "The CRISPR Chronicles: Enter Toolgen"). The Broad list will be the subject of this post.
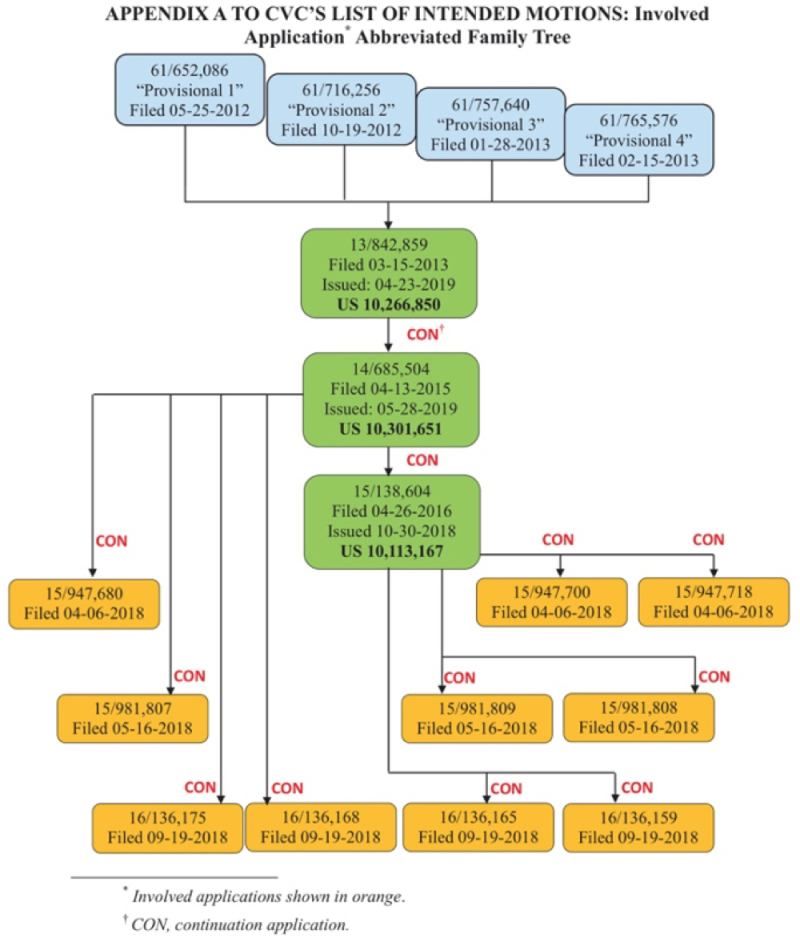
 Broad's list contains six proposed motions and in some respects mirrors the motions filed in on-going Interference No. 106,115 against CVC (see "CRISPR Interference Parties Propose Motions"). Broad Motion No. 1 under 37 C.F.R. §§ 41.121(a)(1)(i) and 41.208(a)(2) asks the Board to substitute the Count in this interference, much as it did in the '115 interference and for essentially the same reasons: "to cover Broad's best proofs, which are directed to CRISPR-Cas9 systems using dual-molecule RNA." In addition, Broad mentions that both parties to this interference contend having invented CRISPR embodiments useful in eukaryotic cells and thus request that priority to this invention be determined by the Board throughout its scope, dual- or single-molecule embodiments. Broad's proposed Count 2 reads as follows:
Broad's list contains six proposed motions and in some respects mirrors the motions filed in on-going Interference No. 106,115 against CVC (see "CRISPR Interference Parties Propose Motions"). Broad Motion No. 1 under 37 C.F.R. §§ 41.121(a)(1)(i) and 41.208(a)(2) asks the Board to substitute the Count in this interference, much as it did in the '115 interference and for essentially the same reasons: "to cover Broad's best proofs, which are directed to CRISPR-Cas9 systems using dual-molecule RNA." In addition, Broad mentions that both parties to this interference contend having invented CRISPR embodiments useful in eukaryotic cells and thus request that priority to this invention be determined by the Board throughout its scope, dual- or single-molecule embodiments. Broad's proposed Count 2 reads as follows:
A CRISPR-Cas9 system, for use in a eukaryotic cell, comprising:
a) a Cas9 or a nucleic acid encoding the Cas9 and
b) an RNA or a nucleic acid encoding the RNA, wherein the RNA is a dual RNA comprising a CRISPR RNA (crRNA) and a trans activating crRNA (tracrRNA) or wherein the RNA is a chimeric RNA comprising a crRNA fused to a tracrRNA,
wherein the crRNA directs the Cas9 to a target sequence in a eukaryotic cell, whereby a site-specific, double-strand break is introduced, or the target sequence is edited.
Broad proposes that, should the Board prefer to include an alternative Count, then Toolgen claim 85 from U.S. Patent Application No. 14/685,510 could be used in the alternative to two Broad claims that Broad has asked in a Contingent Motion (below) be added to this Interference.
Broad Motion No. 2 (contingent) under 37 C.F.R. §§ 41.121(a)(1)(i) and 41.208(a)(2) seeks to add claims 1, 40, and 41 of Broad U.S. Patent Application No. 15/160,71:
Claim 1. An engineered CRISPR-Cas system in a eukaryotic cell having a DNA molecule, the CRISPR-Cas system comprising:
I. a Cas9 or a nucleotide sequence encoding the Cas9, and
II. an RNA or a nucleotide sequence encoding the RNA, the RNA comprising
(a) a first RNA comprising (i) a guide sequence capable of hybridizing to a target sequence of the DNA molecule adjacent to a Protospacer Adjacent Motif (PAM) in the eukaryotic cell, and (ii) a tracr mate sequence, and
(b) a second RNA comprising a tracr sequence capable of hybridizing to the tracr mate sequence,
wherein the guide sequence directs the Cas9 to the target sequence,
whereby the DNA molecule is cleaved or edited in the eukaryotic cell.
and claims 74, 94, and 95 of U.S. Patent Application No. 15/430,260:
Claim 74. A method comprising: introducing into, or expressing in, a eukaryotic cell having a DNA molecule,
(I) a Cas9 protein or a nucleotide sequence encoding the Cas9 protein,
and
(II) an RNA or a nucleotide sequence encoding the RNA, the RNA comprising:
(a) a first RNA comprising a first ribonucleotide sequence and a second ribonucleotide sequence, and
(b) a second RNA, and
wherein, the second RNA forms an RNA duplex with the second ribonucleotide sequence, and
wherein, in the eukaryotic cell, the first ribonucleotide sequence directs the Cas9 protein to a target sequence of the DNA molecule,
whereby the Cas9 cleaves or edits the DNA molecule or alters expression of at least one product of the DNA molecule in the eukaryotic cell.
These claims are in condition for allowance (subject to Broad filing terminal disclaimers), and according to Broad "[b]ased upon communications with the Examiner during a January 26, 2021 interview, Broad understands that claims that are generic to encompass both dual RNA and chimeric RNA embodiments of CRISPR-Cas9 systems for use in and methods to direct site specific binding and cleavage of a target sequence in eukaryotic cells."
Broad Motion No. 3 under 37 C.F.R. §§ 41.121(a)(1)(iii) and 41.208(a)(2) asks the Board to designate claims as not corresponding to Count 1 in the Interference as declared or its Proposed Count 2 should the Board grant Broad's Contingent Motion No. 2. This motion is based on "the limitation to the use of vectors for RNA expression, which is patentably distinct from Count 1 and Proposed Count 2" nor disclosed in Toolgen's application in interference. The patents and claims Broad seeks to exclude on this basis are:
• U.S. Patent No. 8,697,359 – Claims 1-14
• U.S. Patent No. 8,771,945 – Claims 1-25
• U.S. Patent No. 8,795,965 – Claims 1-25
• U.S. Patent No. 8,865,406 – Claims 1-23
• U.S. Patent No. 8,871,445 – Claims 1-25
• U.S. Patent No. 8,889,356 – Claims 1-23
• U.S. Patent No. 8,889,418 – Claims 25-28
• U.S. Patent No. 8,895,308 – Claims 1-24, 29
• U.S. Patent No. 8,906,616 – Claim 24
• U.S. Patent No. 8,932,814 – Claims 1-24
• U.S. Patent No. 8,945,839 – Claims 1-20
• U.S. Patent No. 8,993,233 – Claims 1-43 (all involved claims)
• U.S. Patent No. 8,999,641 – Claims 1-21
• U.S. Patent No. 9,840,713 – Claims 1-14, 37 and 40-41
• U.S. Application No. 14/704,551 – Claims 2 and 4-18 (all involved claims)
• U.S. Application No. 15/330,876 – Claims 1, 16-21, and 30-40 (all involved claims)
This motion asks the Board to designate additional claims as not corresponding to Count 1 or Proposed Count 2 on a different basis, regarding "the limitation to a selected protein, namely, Staphylococcus aureus Cas9":
• U.S. Patent No. 8,865,406 – Claims 1-30 (all involved claims)
• U.S. Patent No. 8,895,308 – Claims 1-30 (all involved claims)
• U.S. Application No. 15/330,876 – Claims 1, 16-21, and 30-40 (all involved claims)
This motion asks the Board to designate additional claims as not corresponding to Count 1 or Proposed Count 2 on yet a different basis, regarding "the limitation that the Cas9 is a chimeric CRISPR enzyme in that it comprises a first fragment and a second fragment, wherein each of the first and second fragments is from a different Cas9 protein":
• U.S. Patent No. 8,889,418 – Claims 1-28 (all involved claims)
This motion asks the Board to designate additional claims as not corresponding to Count 1 or Proposed Count 2 on another different basis, regarding "the limitation to a system for improved localization to the nucleus of a eukaryotic cell, namely, having two or more nuclear localization signals":
• U.S. Patent No. 8,871,445 – Claims 1-30 (all involved claims)
• U.S. Patent No. 8,932,814 – Claims 1-30 (all involved claims)
• U.S. Patent No. 8,993,233 – Claim 7
• U.S. Application No. 14/704,551 – Claims 9-11
• U.S. Application No. 15/330,876 – Claim 34
This motion asks the Board to designate additional claims as not corresponding to Count 1 or Proposed Count 2 on another different basis, regarding "the limitation that the Cas9 comprise one or more mutations, including, but not limited to, whereby as a result of such mutations the Cas9 is a nickase that does not cause cleavage resulting in a double strand break in the target DNA":
• U.S. Patent No. 8,865,406 – Claim 8
• U.S. Patent No. 8,889,356 – Claims 1-30 (all involved claims)
• U.S. Patent No. 8,889,418 – Claims 1-28 (all involved claims)
• U.S. Patent No. 8,932,814 – Claims 1-30 (all involved claims)
• U.S. Patent No. 9,840,713 – Claims 2 and 18-19
• U.S. Application No. 14/704,551 – Claim 5
• U.S. Application No. 15/330,876 – Claim 19-20
This motion asks the Board to designate additional claims as not corresponding to Count 1 or Proposed Count 2 on another different basis, regarding "the limitation that the Cas9 be fused to specified protein domains or includes one or more heterologous domains or includes a functional domain":
• U.S. Patent No. 8,993,233 – Claims 1-43 (all involved claims)
This motion asks the Board to designate additional claims as not corresponding to Count 1 or Proposed Count 2 on another different basis, because "Broad's Involved Claims that are not limited to use of CRISPR Cas system in eukaryotic cells with 'fused' or 'chimeric' RNA" and thus should be removed from the interference should the Board deny Broad's Motion No. 1:
• U.S. Patent No. 8,697,359 – Claims 1-3, 5-10, 12-17 and 19-20
• U.S. Patent No. 8,771,945 – Claims 1-4 and 6-29
• U.S. Patent No. 8,795,965 – Claims 1-30 (all involved claims)
• U.S. Patent No. 8,865,406 – Claims 1-30 (all involved claims)
• U.S. Patent No. 8,871,445 – Claims 1-30 (all involved claims)
• U.S. Patent No. 8,889,356 – Claims 1-30 (all involved claims)
• U.S. Patent No. 8,889,418 – Claims 1-28 (all involved claims)
• U.S. Patent No. 8,895,308 – Claims 1-30 (all involved claims)
• U.S. Patent No. 8,906,616 – Claims 1-4 and 6-30
• U.S. Patent No. 8,932,814 – Claims 1-30 (all involved claims)
• U.S. Patent No. 8,945,839 – Claims 1-28 (all involved claims)
• U.S. Patent No. 8,993,233 – Claims 1-43 (all involved claims)
• U.S. Patent No. 8,999,641 – Claims 1-28 (all involved claims)
• U.S. Patent No. 9,840,713 – Claims 1-7 and 9-41
• U.S. Application No. 14/704,551 – Claims 2 and 4-18 (all involved claims)
These motions are significant, because if the Board grants any or all of them Broad will remove these claims from the risk of having the Board find they are not entitled to priority as to these claims. Specifically, as set forth in Broad's Motion:
Only claims 4, 11, and 18 of U.S. Patent No. 8,697,359; claim 5 of U.S. Patent No. 8,771,945; claim 5 of U.S. Patent No. 8,906,616; claim 8 of U.S. Patent No. 9,840,713, and claims 1, 16-21, and 30-40 of application 15/330,876 should remain designated as corresponding to Count 1.
Broad's Motion No. 4 under 37 C.F.R. §§ 41.121(a)(1)(iii) on the grounds that Toolgen's claims corresponding to the Count are unpatentable. The basis for this motion is that Toolgen is not entitled to the benefit of priority to its earliest-filed application, U.S. Provisional Application No. 61/717,324. Specifically, Broad contends this application does not provide an adequate written description based on several purportedly necessary features of CRISPR in eukaryotic cells (including the timing of the introduction of constructs encoding Cas9 and chimeric RNA; use of Cas9 comprising a single nuclear localization signal at the carboxyl terminus of the protein; and use of a chimeric RNA prepared "exogenous to and before introduction into the eukaryotic cell." In addition, the Broad contends that Toolgen's involved claims are invalid under 35 U.S.C. § 102 and § 103 based on several prior art references, all based on the Board finding Toolgen's involved claims are not entitled to the benefit of priority to its '324 provisional application. (Significantly, because in an interference a party asserting unpatentability of an opponent's claims on prior art grounds is required to show why those references don't invalidate their claims, Broad notes that the Board has granted Broad's patents and applications-in-interference (in the '115 Interference) the benefit of priority to U.S. Provisional Application 61/736,527, having a filing date of December 12, 2012, which antedates all the references Broad asserts against Toolgen's claims in interference.
Broad's Motion No. 5 under 37 C.F.R. §§ 41.121(a)(3) for the Board to keep the parties and the Board apprised of allowance of pending Toolgen applications and to keep the Examiner in each such application apprised on the existence of this Interference.
Finally, Broad's Motion No. 6 under 37 C.F.R. § 41.208(a)(4) seeks judgment based on priority.
The Board having set February 4th as the date for a teleconference with each set of parties jointly, the Board's Order notifying the parties as to which motions they are authorized to file and which have been deferred or denied is expected within the week. During the call the parties were able to inform the Board more specifically of the bases for their motions, provide a proffer if the Board needs additional evidence or information, and provide opposing parties the opportunity to give the Board their views. With the Order on which motions are permitted the Board will also set a briefing schedule. All these events will be the subject of future posts.
* As a reminder, an interference proceeds in two stages. The first stage involves the parties presenting motions that can modify the count, have certain claims declared outside the scope of the count (or vice versa), seek to establish an earlier priority date, and ask for a finding that their opponents' claims are invalid under any of the provisions of the patent statute. If these motions are not decided in a way that would disqualify one or both parties, then the interference will move to a second stage, where in each Interference the Junior Party (Broad, in the '126 Interference and CVC in the '127 Interference) will present its proofs of conception and reduction to practice and the Senior Party Toolgen will be permitted to oppose and/or present its own priority evidence. The Senior Party is under no obligation to present proofs earlier than its earliest filing date unless the Junior Party evinces evidence of (at least) earlier conception. In practice, the parties can both be expected to submit their priority evidence.
 Last month, the Intellectual Property Owners Association (IPO) and Harrity Analytics announced the release of the 38th annual list of the top 300 organizations receiving U.S. utility patents (see "IPO & Harrity Analytics Release List of Top 300 Patent Holders for 2019"). Patent Docs readers may recall that the U.S. Patent and Trademark Office stopped releasing its annual list of top patent recipients in 2006 in order to "discourag[e] any perception that we believe more is better."
Last month, the Intellectual Property Owners Association (IPO) and Harrity Analytics announced the release of the 38th annual list of the top 300 organizations receiving U.S. utility patents (see "IPO & Harrity Analytics Release List of Top 300 Patent Holders for 2019"). Patent Docs readers may recall that the U.S. Patent and Trademark Office stopped releasing its annual list of top patent recipients in 2006 in order to "discourag[e] any perception that we believe more is better." As with the 2019 report, the 2020 report was compiled in collaboration with Harrity Analytics, based on data obtained from the U.S. Patent and Trademark Office. The IPO and Harrity Analytics noted that patents reported on the 2020 list are utility patents granted during the 2020 calendar year that listed the organization or a subsidiary as the owner on the printed patent document. Harrity Analytics then used organization and subsidiary data provided to two independent third-party data providers and reviewed over 200,000 assignments to attribute ownership to the correct organizations, where applicable. The report notes that patents that were granted to two or more organizations jointly are attributed to each organization separately, and that companies for which patents were granted to subsidiaries are combined with patents granted to the parent.
As with the 2019 report, the 2020 report was compiled in collaboration with Harrity Analytics, based on data obtained from the U.S. Patent and Trademark Office. The IPO and Harrity Analytics noted that patents reported on the 2020 list are utility patents granted during the 2020 calendar year that listed the organization or a subsidiary as the owner on the printed patent document. Harrity Analytics then used organization and subsidiary data provided to two independent third-party data providers and reviewed over 200,000 assignments to attribute ownership to the correct organizations, where applicable. The report notes that patents that were granted to two or more organizations jointly are attributed to each organization separately, and that companies for which patents were granted to subsidiaries are combined with patents granted to the parent.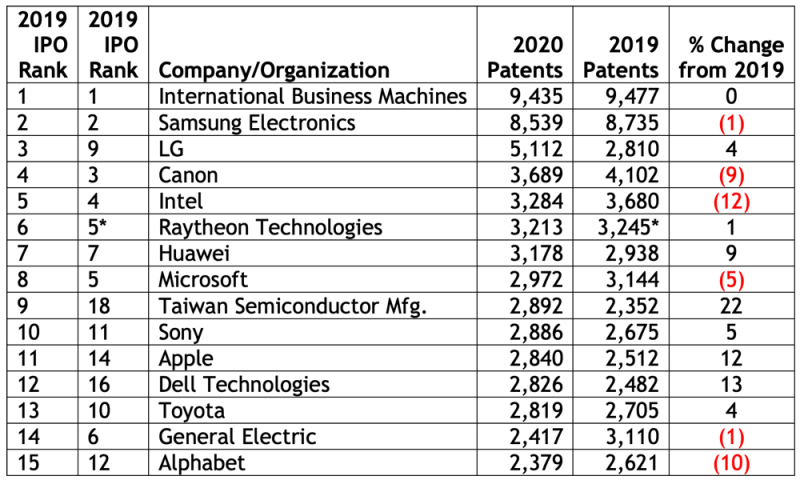 * Merger of United Technologies and Raytheon.
* Merger of United Technologies and Raytheon.