By Kevin E. Noonan –
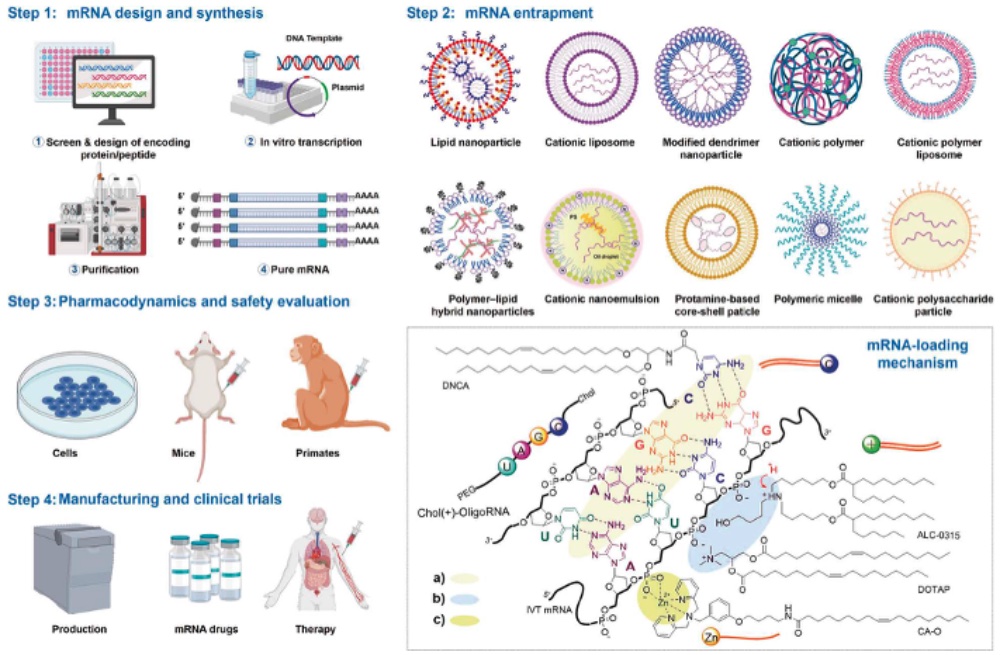
A consequence of the gradual decline in COVID infections worldwide has been a less gradual increase in patent litigation by the many entities having patent rights for the vaccines or components of them. This is particularly true of the so-called “mRNA” vaccines, which have been the subject of development by many companies over the past thirty to forty years. The subject matter of these patents have included the mRNA “payload” itself, which can comprise structural modifications such as containing various pseudouridine residues and percentage substitution in the molecule, as well as the genetic structure of the mRNA itself. Perhaps even more complexity can be found in the protective “covering” of the mRNA (which is notoriously labile without such protections), examples of which are illustrated in the upper righthand quadrant of this drawing:
The Moderna vaccine formulation (relevant to Plaintiff’s complaint for infringement) is set forth in the chart below:
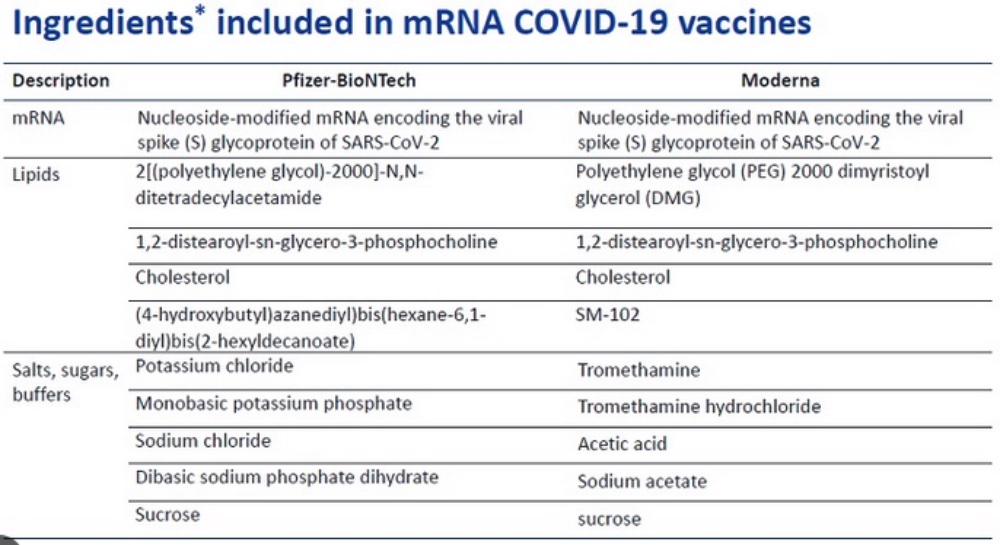
In their complaint (Arbutus Biopharma Corp. & Genevant Biosciences GmbH v. Moderna, U.S. District Court for the District of Delaware, No. 1:22-cv-00252), Plaintiffs Arbutus and Genevant alleged that Moderna infringed claims of U.S. Patent Nos. 8,058,069, 8,492,359, 8,822,668, 9,364,435, 9,504,651, and 11,141,378. Arbutus/Genevant asserted these patents were infringed by Moderna for incorporating into its Spikemax® COVID vaccine lipid nanoparticles (LNP) comprising an ionizable cationic lipid (designated SM-102):
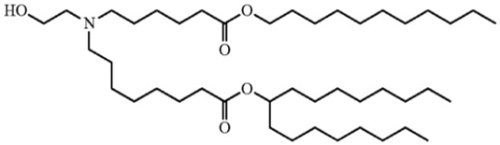
a phospholipid (DSPC); cholesterol; and a conjugated lipid that inhibits aggregation of particles (a PEG-lipid conjugate), with certain advantageous percentages of the components and with encapsulated mRNA. Claim 1 from each patent is set forth in this table:

In press releases from the parties, it was announced that Moderna had settled, and that the settlement terms included an immediate $950 million payment (to be paid in July, 2026) that settled all infringement damages; and a provision for a further $1.3 billion in damages depending on the outcome of Moderna’s appeal regarding its allegations that the Federal government was responsible for these monies under the provisions of 28 U.S.C. § 1498(a). In this regard a district court denied Moderna’s partial motion to dismiss (on jurisdictional grounds), holding that Moderna was not entitled to Section 1498 protection because the accused infringing vaccines were not limited to government use, despite the Department of Justice filing a brief to the contrary. This court in this case on February 2nd held that production of Moderna’s vaccine under “Operation Warp Speed” did not override Plaintiffs’ patent rights. Arbutus/Genevant retain the rights to pursue claims before the Court of Federal Claims should Moderna prevail on its Section 1498 appeal, and Moderna will pay $1.3 billion to Arbutus/Genevant should they not. Moderna also received a global nonexclusive license to patented technology for SM-102-containing mRNA vaccines for infectious diseases (specifically SPIKEVAX, mNEXSPIKE and mRESVIA (according to Arbutus’s Form 8-K filing with the Securities and Exchange Commission on March 5th), and an agreement not to sue for “certain” (undisclosed) patents on Moderna’s products using the technology; it is unclear whether Moderna vaccines against diseases other than infectious disease would fall within the scope of this license (for example, Moderna’s anticancer vaccines, which have recently had positive clinical trial results; see “Merck-Moderna cancer vaccine sustains 49% melanoma risk reduction at 5 years“). See “Roivant Sciences Ltd. (ROIV) Discusses $2.25 Billion Global Settlement Resolving COVID Vaccine Patent Litigation Transcript” for further details. Moderna also agreed for the District Court to enter judgment that four of Arbutus/Genevant’s asserted patents were not invalid. Finally, the agreement (according to the SEC disclosure) encompasses international disputes between the parties over this technology. At present, these include lawsuits in Canada, Japan, and Switzerland and two actions before the Unified Patent Court (UPC), which seek relief for 28 countries including France, Germany, Greece, Italy, Ireland, and Spain.
For further context, Moderna has received $48 billion in past global vaccine sales (according to Reuters).
These plaintiffs filed a similar lawsuit against Pfizer/BioNTech over the LNP technology in their vaccine in 2023. That lawsuit is still ongoing.
Moderna has separately sued rivals Pfizer and BioNTech for infringing patents related to mRNA technology. And BioNTech countersued Moderna in February, arguing Moderna’s next-generation COVID-19 shot, MNEXSPIKE, infringes its U.S. Patent No. 12,133,899.
Earlier this month, Bayer filed suit separately against Moderna, Inc.; Moderna US, Inc.; and ModernaTx Inc.; Pfizer Inc.; BioNTech SE; BioNTech Mfg. GmbH; and BioNTech US Inc.; and Johnson & Johnson; Janssen Pharmaceutica NV; Janssen Pharmaceuticals Inc.; Janssen Biotech Inc.; and Janssen Vaccines & Prevention N.V. with respect to U.S. Patent No. 7,741,118, which reduces the number of polyadenylation signals (ATTTA) in an mRNA payload and as a result improves translation efficiency.
It appears unlikely that these judicial contretemps will end any time soon.

Leave a comment