By Kevin E. Noonan –

One of the paradoxically beneficial consequences of the COVID pandemic was that even without the compulsory aspects of the vaccine patent waiver proposed by certain WTO member states (Brazil, India, and South Africa in particular) vaccine makers, especially Moderna and BioNTech/Pfizer foreswore exercising their patent rights in favor of facilitating as widespread a distribution of vaccine as possible. This behavior was particularly significant in light of the long term investment in funding and scientific effort these technologies represented for these and other companies.

That was then.
On February 19th, BioNTech filed a complaint in the U.S. District Court for the District of Delaware asserting that Moderna and its subsidiaries were infringing claims of BioNTech’s U.S. Patent No. 12,133,899, wherein claim 1 is representative:
1. A pharmaceutical composition comprising an RNA that:
(i) includes modified uridines in place of all uridines [which in dependent claims are each N1-methyl pseudouridine], and
(ii) comprises a nucleotide sequence that encodes a polypeptide, wherein the polypeptide comprises:
(a) one or more fragments of a SARS-CoV-2 Spike (S) protein, wherein one of the fragments comprises a receptor binding domain (RBD),
(b) a secretory signal [which in dependent claims are from the SARS-CoV-2 S protein]; and
(c) one or more additional domains selected from a trimerization domain [which in dependent claims are from T4 fibritin protein], a transmembrane domain [which in dependent claims are from the SARS-CoV-2 S protein], and a combination thereof;
wherein the RBD is linked to one of the additional domains via a linker, and wherein the linker comprises a GS [Glycine-Serine] linker.
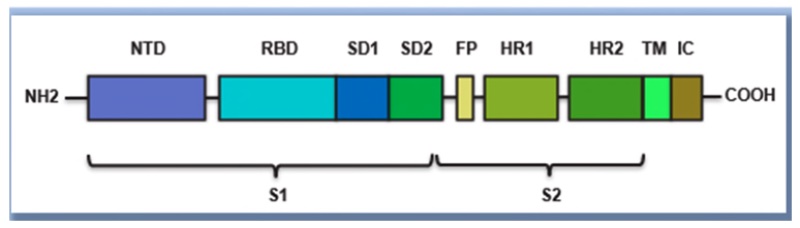
BioNTech accuses Moderna of using its mRNA construct comprising these components in its mNEXSPIKE® vaccine against COVID (SARS-CoV-2). The claimed mRNA comprises a “streamlined vaccine design” according to the complaint, which triggers “an equally strong immune response to that of the complete spike protein” despite comprising only select domains thereof, specifically, a receptor binding domain, a secretory signal, a transmembrane domain, all from the SARS-CoV-s Spike protein, and a trimerization domain from the heterologous T4 fibritin protein; the full-length Spike protein structure is illustrated in the complaint as:
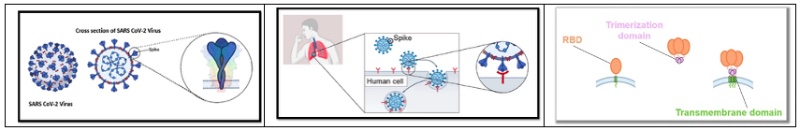
According to BioNTech, the encoded protein mimics the full-length Spike protein biology, as illustrated by these drawings:
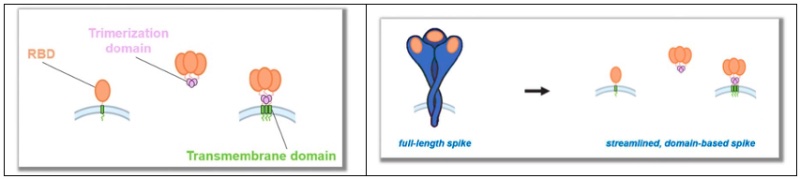
And the structure of the individual components and the “streamlined, domain-based Spike” here:
This vaccine can be given to patients at lower dosages and is more stable during storage and transport, according to the complaint. The complaint also highlights how Moderna itself lionizes its new (according to BioNTech, copied) form of its COVID vaccine, particularly with regard to being able to administer a lower dose and the advantages of using lower dosages, with concomitant improvements in profitability.
The basis for BioNTech’s infringement allegations are these illustrations of Moderna’s mNEXSPIKE® vaccine:
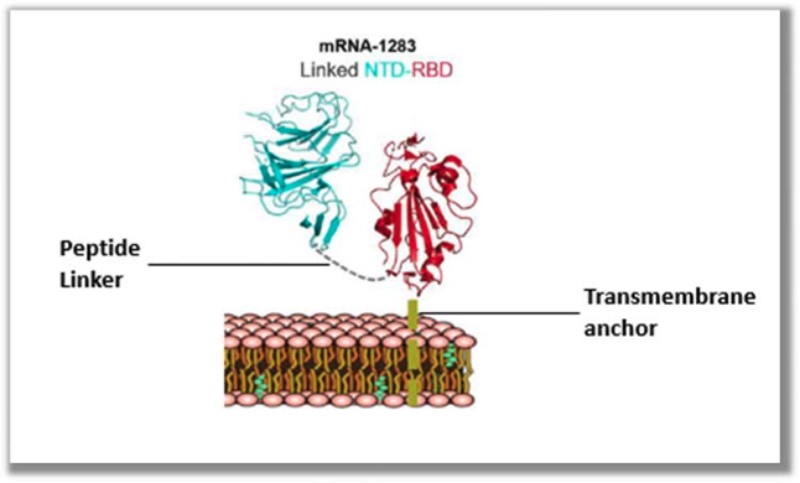
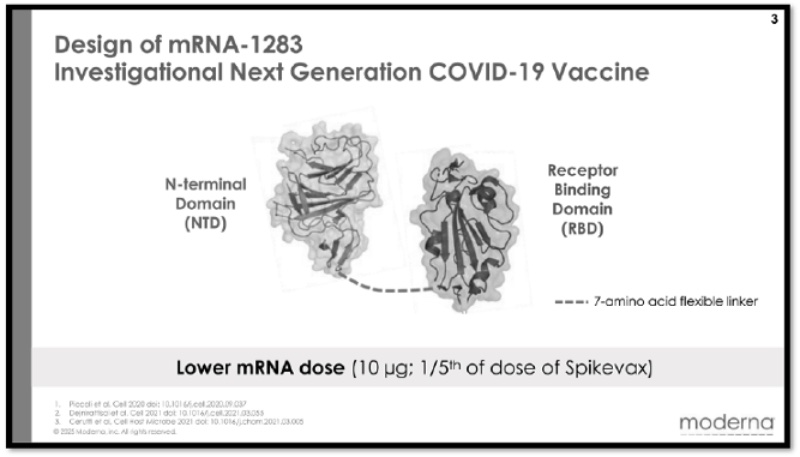
accompanied by descriptions of the components in the label (including N1-methyl-pseudoruridine substitution, and the presence of a secretory signal, a receptor binding domain, and a transmembrane domain joined with a peptide linker).
In its prayer for relief, BioNTech asks for money damages for literal, induced, or contributory infringement and that the infringement be adjudged to be willful, as well as having the court find this to be an exceptional case for which BioNTech will be entitled to attorneys’ fees, costs, and expenses incurred in connection with this action. Significantly, in this action BioNTech maintains the practice not to ask for an injunction.

Leave a comment